 MYKOLA POKHODZHAY / ISTOCK / GETTY IMAGES PLUS
MYKOLA POKHODZHAY / ISTOCK / GETTY IMAGES PLUS
Evidence-Based Approach to Avoid Postop Sensitivity in Adhesive Dentistry
A literature review discussing potential clinical factors contributing to postoperative sensitivity after placing adhesive restorations.
Despite developing trends in restorative procedures and improvements in materials, dental postoperative sensitivity (POS) remains a prevalent issue for clinicians performing direct adhesive restorations. The risk of developing POS can be as high as 20% to 40%,1–3 making it a significant complication. Historically, the literature assesses POS as a secondary outcome to material performance. Consequently, there is a lack of controlled research identifying links between clinical techniques and the incidence of POS. This literature review aims to discuss potential factors contributing to POS after placement of adhesive restorations, as well as measures to minimize its occurrence. Treatment and prevention of POS is highly case specific; thus, clinicians may wish to consider a combination of techniques while initiating restorative treatment to avoid this phenomenon.
Pain in a tooth linked with chewing or sensitivity to hot, cold and/or sweet stimuli that occurs a week or longer after restoration is characterized as POS.4 Every patient’s spectrum of sensitivity is different, but it is usually immediate and transient. With repeated treatment of the tooth, deeper caries lesions,5,6 or complex lesion configurations,1 POS is probable, and the tooth will need to be monitored over time.
In terms of stimulus, temperature sensitivity (especially to cold) and masticatory sensitivity have been the most commonly reported manifestations of POS. Sensitivity to cold temperatures was prevalent mainly in Class II restorations, but the sensation can be expected to fade in small- to moderate-sized restorations after a week.1 Sensitivity to biting forces can occur when a portion of the direct restoration may have remained “high” or is pressing harder on the opposite tooth, but the sensation usually subsides with occlusal adjustment. Pain during clenching can indicate hyperocclusion; however, pain during chewing — otherwise called occlusal loading sensitivity — results from fluid accumulation within gaps between the restoration and dentin. As the tooth and restoration deform during mastication, fluid flows down the dentinal tubules, inducing sensitivity.7
In rare situations, POS lingers, and masticatory or temperature sensitivity never subsides, despite time and occlusal adjustment. This happens commonly in deeper caries lesions with minimal remaining dentin thickness or a tooth receiving treatment for the second or third time.6,8 Substantial and repeated restorative therapies can lead to persistent sensitivity or pulpal irritation, resulting in the affected tooth needing root canal treatment.6,8
CAUSES OF POSTOPERATIVE SENSITIVITY
The phenomenon of pain or sensitivity elicited in dentin through dentinal fluid movement (explained by the widely accepted hydrodynamic theory) provides insights into how dentin and its components may react to different stimuli introduced during a restorative procedure.9 According to this theory, various factors can cause dentinal fluid movement, including dentin dehydration, temperature variations, and penetration of chemical or bacterial agents.9
Restoration-derived sensitivity is dependent on microleakage, polymerization shrinkage, and C factor (the ratio of bonded over unbonded surfaces).10 Microleakage around restorations can result from marginal gaps or voids, degradation of the bonding or restorative material, dissolution of smear layers, and varying coefficients of thermal expansion for restorations.3 Polymerization shrinkage can cause internal stress and deformation in surrounding tooth structure, resulting in poor marginal adaptation that increases the risk of POS.11
CARIES EXCAVATION
Increased caries depth is associated with a greater risk of POS due to an increase in the number and size of dentinal tubules subject to pulpal irritation or microleakage.5 Dry cutting dentin and the use of blunt burs can generate frictional heat, contributing to POS.10,12 Clinicians are advised to employ intermittent cutting strategies and ensure adequate water irrigation during cavity preparation and replace worn-out burs to improve cutting efficiency and minimize heat-induced pulpal damage.10
ETCHING TIME AND STRATEGY
Prolonged etching time (> 15 seconds) can result in morphological changes in the dentin surface, such as widened tubule openings, increased tubule diameter, and greater smear layer dissolution, with corresponding deeper dentin demineralization.13 This phenomenon contributes positively to mechanical interlocking and resin tag formation, but negatively impacts hybridization. Accidental over-etching can create a demineralized dentin depth of more than 5 microns, which may exceed the penetration potential of the adhesive. This results in a weaker hybrid layer formation, with gaps prompting nanoleakage, thereby increasing susceptibility to POS.14 Prolonged acid exposure can also denature fragile collagen fibers, resulting in increased dentin permeability.14
There is abundant literature on choosing between self-etch and total-etch techniques to avoid POS, with varying and often contradictory findings. Some studies suggest that self-etch strategies are more effective in curbing immediate sensitivity within 24 hours in deeper caries lesions than the total-etch technique.15 However, the current evidence shows no difference in POS with either method.16
ADHESIVE APPLICATION
Pooling of adhesive from a lack of air thinning can lead to improper solvent evaporation and incomplete polymerization.17 Similarly, incomplete coating of adhesive results in micro/nanoleakage from a compromised tooth-restoration adhesive interface, triggering POS.10,17 Therefore, the adhesive application technique demands closely following the manufacturer’s instructions for use.
Dentin moisture levels can affect the occurrence of POS, as the dentin collagen matrix collapses on excessive drying, which results in incomplete monomer penetration and resulting voids.17 Consequently, amidst contradictory clinical reports,18 water or ethanol wet bonding techniques have been favored over the years.19
INCREMENTAL VERSUS BULK FILLING
Traditionally, the incremental filling technique — in which composite resin is placed in increments of 2 mm maximum thickness — was aimed at ensuring adequate polymerization, improving marginal adaptation, and reducing shrinkage by lowering the C-factor.20 Conversely, incremental filling creates a risk of voids between layers, marginal gap formation, and contamination; it is also time-consuming.21 For faster and simpler procedures, the development of bulk-fill resins allowed increments of up to 4 to 5 mm thickness. Contemporary bulk-fill composites have an adequate degree of conversion, low volumetric shrinkage, and a high depth of cure, even when used at a 4 mm thickness.22 Though bulk-filling has its benefits, studies have reported that bulk placement of conventional non-bulk-fill composites in deep caries lesions can increase cuspal deflection and induce stress at the adhesive interface, which clinically manifests as POS.23
When comparing both techniques, Costa et al3 demonstrated the overall risk of immediate POS was not affected by the restorative technique (incremental or bulk). The clinical outcomes of POS with incremental and bulk-fill resins in direct posterior restorations were similar, suggesting that choice of technique may be inconsequential in the development of POS.24
CURING
Under-curing leaves bonding resin only partly polymerized, which can induce sensitivity.12 If curing devices do not produce recommended light intensity levels (irradiance output: 300 to 400 mW/cm2), they cause insufficient polymerization, resulting in the harmful release of unreactive monomer and camphorquinone into the pulp.10,25 It is essential to ensure curing devices are powerful enough, with the correct tip diameter (around 8 mm) for light to penetrate the entire distance to the cavity preparation base.12,25 Furthermore, using a soft-start curing mode induces slow initial polymerization and can reduce shrinkage within the restoration, potentially reducing POS occurrence.26 Light-curing units must be stabilized intraorally with the operator’s fingers and oriented perpendicular to the tooth, starting at a distance of 1 cm from the restoration and gradually bringing them closer for successful curing.25
FINISHING AND POLISHING
Finishing and polishing should be limited to minor contour corrections and superficial smoothening, as this will help minimize issues with POS. Placing excess composite material, followed by aggressive finishing, can alter the resin matrix, disturb the post-irradiation phase of polymerization, and remove the highly polymerized superficial layer.10 Reportedly, finishing 24 hours after placement of microfilled composite restorations on the dentin margin with diamond burs under wet conditions produces significantly lower microleakage, thus minimizing POS frequency.27
PREVENTING POSTOPERATIVE SENSITIVITY
A tooth’s preoperative conditions (e.g., size, depth and location of lesions, anatomical differences, microcracks, preexisting hypersensitivity, enamel/dentin defects, or pulpal health) can trigger POS.10,12 While clinicians cannot control these factors directly, they can prevent POS by paying attention to restorative strategies and minimizing technique errors when placing direct composite restorations. Furthermore, operators can reduce POS occurrence by updating their skills and knowledge regarding a specific material’s characteristics, handling techniques, indications and limitations.
REDUCING TECHNIQUE-RELATED ERRORS
Preoperatively, clinicians are advised to inspect the tooth for other factors that can contribute to sensitivity — such as abrasions, cervical dentin exposure, pulpal inflammation, enamel/dentin defects, or similar concerns — before initiating restorative treatment. Based on the tooth’s clinical condition(s), deploying various strategies can minimize the risk of POS at each stage, as described in Table 1. Postoperatively, incomplete tooth fractures that can mimic POS may develop due to extended caries preparations; if present, these must be appropriately diagnosed and addressed. If POS persists after restorative treatment, it may be necessary to replace the restoration. 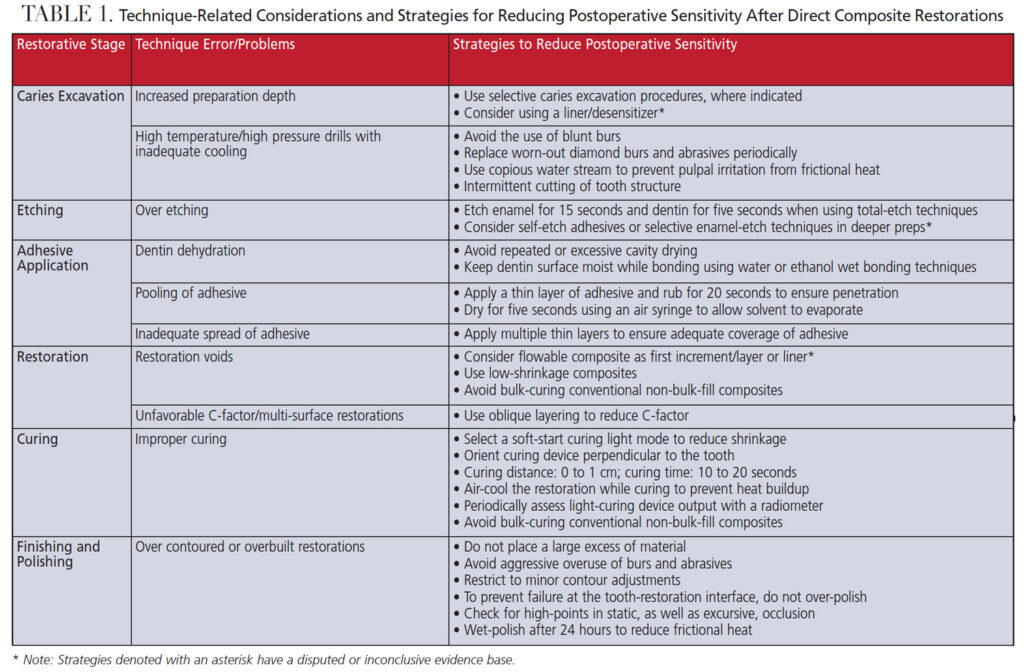
MATERIAL CONSIDERATIONS
In adhesive dentistry, the choice of adhesive material is not as consequential as the technique employed to minimize the risk of POS.16 Operator skill and experience are related to POS outcomes.28 Inexperienced practitioners can start by using simple single-step techniques for adhesion.28 More advanced operators may be comfortable with the highly technique-sensitive multistep adhesive systems that can help reduce POS incidence.28
Given their prior success in curbing preoperative dentin exposure-related hypersensitivity, using dentin desensitizers in deeper caries preparations prior to acid etching has been contemplated as a strategy to reduce POS.12,29 Examples of commonly used desensitizers include glutaraldehyde/hydroxyethylmethacrylate, chlorhexidine, fluoride-based agents and potassium oxalate. While various desensitizers do not significantly differ in terms of their efficacy in reducing dentinal hypersensitivity, clinical trials noted no benefits with these agents when used under direct or indirect restorations.2,30,31 Furthermore, most desensitizers, including oxalate and fluoride-based agents, compromise an adhesive’s bond strength to dentin.32
Liners, such as resin modified glass ionomer and calcium hydroxide, have been investigated regarding their effect in reducing POS.33 A Cochrane review indicated that liners offer no additional benefits in mitigating POS, unless intended for a therapeutic effect.33 However, it concluded there was insufficient data and a high risk of bias among studies to make a robust clinical recommendation.33 Flowable composites, due to their low viscosity and elastic modulus, theoretically result in better marginal adaptation and reduced microleakage if used as the first increment.20 Yet clinical trials saw little value in terms of microleakage or clinical performance to reduce POS.34 Being less susceptible to polymerization shrinkage, low-shrinkage resins and bulk-fill ormocers are hypothesized to elicit less POS than conventional composites; however, a meta-analysis determined this effect is statistically insignificant and both are clinically comparable.35
Interestingly, low-level diode laser irradiation has the potential to reduce POS when used before resin placement in restorations.36 Similarly, air abrading the preparation using a 50-micron aluminum oxide microetcher before placing the restoration reduces microleakage-related POS.37 However, a larger evidence base with more robust clinical trials is required to validate these strategies for preventing POS.
CONCLUSION
There is no one-size-fits-all solution to combat POS. Since various tooth-related, material-related, and technique-related factors contribute to POS, clinicians are encouraged to consider strategies to mitigate the risk before initiating treatment. Practitioners should carefully select a combination of measures, materials and techniques in each clinical situation to personalize treatment for the patient. In general, clinicians would benefit from drawing on research-supported recommendations for each step of restorative treatment, and establish a standard clinical workflow based on their past experiences of successful treatment to ensure more predictable outcomes and minimize the incidence of POS.
References
- Briso AL, Mestrener SR, Delício G, et al. Clinical assessment of postoperative sensitivity in posterior composite restorations. Oper Dent. 2007;32:421–426.
- de Oliveira ILM, Hanzen TA, de Paula AM, et al. Postoperative sensitivity in posterior resin composite restorations with prior application of a glutaraldehyde-based desensitizing solution: A randomized clinical trial. J Dent. 2021;117:103918.
- Costa T, Rezende M, Sakamoto A, et al. Influence of adhesive type and placement technique on postoperative sensitivity in posterior composite restorations. Oper Dent. 2017;42:143–154.
- Berkowitz G, Spielman H, Matthews A, et al. Postoperative hypersensitivity and its relationship to preparation variables in Class I resin-based composite restorations: findings from the Practitioners Engaged in Applied Research and Learning (PEARL) Network. Part 1. Compend Contin Educ Dent. 2013;34:e44–e52.
- Auschill TM, Koch CA, Wolkewitz M, Hellwig E, Arweiler NB. Occurrence and causing stimuli of postoperative sensitivity in composite restorations. Oper Dent. 2009;34:3–10.
- Abbas B, Gulfam F, Anwar FS, Zara B, Aamer S, Zafar S. Influence of cavity depth and liner on postoperative hypersensitivity in posterior composite restorations. Med Forum. 2021;32:121–125.
- Hirata K, Nakashima M, Sekine I, Mukouyama Y, Kimura K. Dentinal fluid movement associated with loading of restorations. J Dent Res. 1991;70:975–978.
- Hayashi M, Wilson NH. Failure risk of posterior composites with post-operative sensitivity. Oper Dent. 2003;28:681–688.
- Brannstrom M. The hydrodynamic theory of dentinal pain: sensation in preparations, caries, and the dentinal crack syndrome. J Endod. 1986;12:453–457.
- Porto IC. Postoperative sensitivity in direct resin composite restorations: clinical practice guidelines. IJRD. 2012;1:1–2.
- Soares CJ, Faria-E-Silva AL, Rodrigues MP, et al. Polymerization shrinkage stress of composite resins and resin cements — What do we need to know? Braz Oral Res. 2017;31(Suppl 1):e62.
- Sabbagh J, Fahd JC, McConnell RJ. Post-operative sensitivity and posterior composite resin restorations: a review. Dental Update. 2018;45:207–213.
- Brajdić D, Krznarić OM, Azinović Z, Macan D, Baranović M. Influence of different etching times on dentin surface morphology. Coll Antropol. 2008;32:893–900.
- Cardenas AFM, Siqueira FSF, Bandeca MC, et al. Impact of pH and application time of meta-phosphoric acid on resin-enamel and resin-dentin bonding. J Mech Behav Biomed Mater. 2018;78:352–361.
- Yousaf A, Aman N, Manzoor MA, Shah JA, Rasheed D. Postoperative sensitivity of self etch versus total etch adhesive. J Coll Physicians Surg Pak. 2014;24:383–386.
- Reis A, Dourado Loguercio A, Schroeder M, Luque-Martinez I, Masterson D, Cople Maia L. Does the adhesive strategy influence the post-operative sensitivity in adult patients with posterior resin composite restorations? A systematic review and meta-analysis. Dent Mater. 2015;31:1052–1067.
- Hayashi M. Adhesive dentistry: understanding the science and achieving clinical success. Dent Clin North Am. 2020;64:633–643.
- Castro AS, Maran BM, Gutiérrez MF, et al. Dentin moisture does not influence postoperative sensitivity in posterior restorations: A double-blind randomized clinical trial. Am J Dent. 2020;33:206–212.
- Pashley DH, Tay FR, Carvalho RM, et al. From dry bonding to water-wet bonding to ethanol-wet bonding. A review of the interactions between dentin matrix and solvated resins using a macromodel of the hybrid layer. Am J Dent. 2007;20:7–20.
- Ferracane JL, Lawson NC. Probing the hierarchy of evidence to identify the best strategy for placing class II dental composite restorations using current materials. J Esthet Restor Dent. 2021;33:39–50.
- Soares CJ, Rosatto C, Carvalho VF, Bicalho AA, Henriques J, Faria-E-Silva AL. Radiopacity and porosity of bulk-fill and conventional composite posterior restorations — digital X-ray analysis. Oper Dent. 2017;42:616–625.
- Van Ende A, De Munck J, Lise DP, Van Meerbeek B. Bulk-fill composites: a review of the current literature. J Adhes Dent. 2017;19:95–109.
- Vinagre A, Ramos J, Alves S, Messias A, Alberto N, Nogueira R. Cuspal displacement induced by bulk fill resin composite polymerization: biomechanical evaluation using fiber Bragg grating sensors. Int J Biomater. 2016;2016:7134283.
- Arbildo-Vega HI, Lapinska B, Panda S, Lamas-Lara C, Khan AS, Lukomska-Szymanska M. Clinical effectiveness of bulk-fill and conventional resin composite restorations: systematic review and meta-analysis. Polymers (Basel). 2020;12:1786.
- Price RB, Dickie D, Strassler HE. Guidelines for successful light-curing. Inside Dental Assisting. 2014;11(4):30–38.
- Alomari Q, Omar R, Akpata E. Effect of LED curing modes on postoperative sensitivity after Class II resin composite restorations. J Adhes Dent. 2007;9:477–481.
- Lopes GC, Franke M, Maia HP. Effect of finishing time and techniques on marginal sealing ability of two composite restorative materials. J Prosthet Dent. 2002;88:32–36.
- Sancakli HS, Yildiz E, Bayrak I, Ozel S. Effect of different adhesive strategies on the post-operative sensitivity of class I composite restorations. Eur J Dent. 2014;8:15–22.
- Mehta D, Gowda VS, Santosh A, Finger WJ, Sasaki K. Randomized controlled clinical trial on the efficacy of dentin desensitizing agents. Acta Odontol Scand. 2014;72:936–941.
- Chermont AB, Carneiro KK, Lobato MF, et al. Clinical evaluation of postoperative sensitivity using self-etching adhesives containing glutaraldehyde. Braz Oral Res. 2010;24:349–354.
- Eyüboğlu GB, Naiboğlu P. Clinical efficacy of different dentin desensitizers. Oper Dent. 2020;45:E317–E333.
- Li J, Hua F, Xu P, Huang C, Yang H. Effects of desensitizers on adhesive-dentin bond strength: a systematic review and meta-analysis. J Adhes Dent. 2021;23:7–19.
- Schenkel AB, Veitz-Keenan A. Dental cavity liners for class I and class II resin-based composite restorations. Cochrane Database Syst Rev. 2019;3:CD010526.
- Boruziniat A, Gharaee S, Sarraf Shirazi A, Majidinia S, Vatanpour M. Evaluation of the efficacy of flowable composite as lining material on microleakage of composite resin restorations: A systematic review and meta-analysis. Quintessence Int. 2016;47:93–101.
- Kruly PC, Giannini M, Pascotto RC, et al. Meta-analysis of the clinical behavior of posterior direct resin restorations: Low polymerization shrinkage resin in comparison to methacrylate composite resin. PLoS One. 2018;13:e0191942.
- Rezaei-Soufi L, Taheri M, Fekrazadas R, Farhadian M. Effect of 940 nm laser diode irradiation prior to bonding procedure on postoperative sensitivity following class II composite restorations: a split-mouth randomized clinical trial. Lasers Med Sci. 2021;36:1109–1116.
- Arora A, Acharya SR, Vidya SM, Sharma P. A comparative evaluation of dentinal hypersensitivity and microleakage associated with composite restorations in cavities preconditioned with air abrasion — An ex vivo study. Contemp Clin Dent. 2012;3:306–313.
From Decisions in Dentistry. July 2022;8(7)16-19.


