 ELENATHEWISE/ISTOCK/GETTY IMAGES PLUS
ELENATHEWISE/ISTOCK/GETTY IMAGES PLUS
Clinical Considerations for Drug Interactions
The provision of safe care requires dental teams to carefully assess the possibility of adverse drug interactions.
This course was published in the March 2020 issue and expires March 2023. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the incidence of polypharmacy in the United States and its potential impact on adverse drug interactions and clinical care.
- Explain the stages of pharmacokinetics, and how drugs can react with other drugs — as well as food and vitamin/mineral/herbal supplements — during each stage.
- List clinical strategies for assessing and minimizing the risk of adverse drug interactions.
The polypharmaceutical nature of modern health care, with an estimated 29% of Americans taking five or more medications on a regular basis,1 requires medical and dental professionals to have a thorough understanding of possible drug interactions. Drugs can interact with each other, as well as with herbal supplements, vitamin and mineral supplements, and even various foods.2 Understanding the many complex, underlying mechanisms of these interactions and resulting intricate drug relationships is crucial in preventing adverse drug reactions and potentially life-threatening emergencies.2 While the number and therapeutic categories of drugs prescribed in the dental setting is limited compared to medicine, a thorough review of the patient’s drug history and evaluation for potential adverse reactions — or reduced effect as a result of drug interaction — is required.3,4
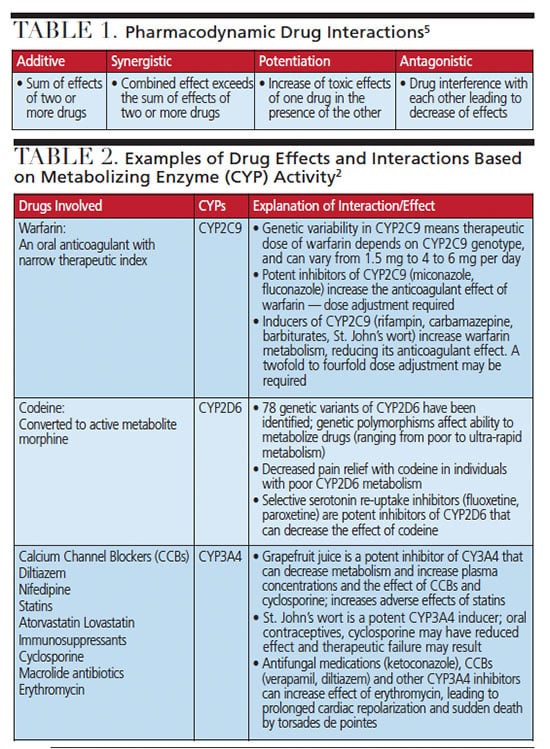
Drugs interact with other drugs, food and supplements at each stage of their pharmacokinetics (absorption, distribution, metabolism and elimination — or ADME), and at the sites of action with their extra- and intracellular receptors (pharmacodynamics).2,3,5 Functionally, these intricate drug relationships can be explained as additive, synergistic, potentiation or antagonistic reactions (Table 1).5 A review of the mechanisms of pharmacodynamic and pharmacokinetic interactions at each stage of a drug’s “life” in the human body will assist oral health professionals in understanding the elaborate processes behind clinically important drug interactions.
INTERACTIONS OF ABSORPTION
The first pharmacokinetic step — absorption — occurs in the stomach and small intestine and can be affected by numerous factors, including gastric pH, activity of the mucosal transport proteins (such as P-glycoprotein), gut microbial flora and gastrointestinal motility.3,4,6 Consequently, drugs, food or herbal supplements that influence these factors can change the rate or, more importantly, extent of drug absorption, affecting its bioavailability and clinical effectiveness. A well-known example of the effect of gastric pH involves its absorption of the antifungal agent ketoconazole, which requires a very acidic environment. Antacids, proton-pump inhibitors (e.g., esomeprazole) and histamine-2 receptor blockers (cimetidine) that increase pH will reduce the absorption of ketoconazole and, thus, its clinical effectiveness.3,4
The bioavailability of some antibiotics, including ciprofloxacin, azithromycin and tetracycline, can be reduced by as much as 43% due to forming of poorly absorbed complexes with various cations (Al3+, Ca2+, Bi2+, Fe2+, Mg2+, Zn2+) contained in certain foods, antacids, and vitamin and mineral supplements.3,4,6 Patients should be cautioned about the potential interactions of drug absorption and advised to carefully review the prescribing information.
INTERACTIONS OF DISTRIBUTION
Once absorbed into the bloodstream, many drugs are distributed through the body either as available free drug or bound reversibly to plasma proteins, mainly albumin. The percentage bound to protein, and therefore not available at the intended site of action, depends on the drug concentration and its affinity for that plasma protein. Drug interactions may occur when protein binding sites, which are limited in number, are saturated to capacity. When two or more drugs compete for the same binding sites, with one drug displacing another, it increases its availability as a free active drug.5 With many drugs, a small increased percentage may have no clinically significant effect.4 However, displacement is problematic when a highly bound drug with a narrow therapeutic index, such as warfarin (which is up to 99% protein-bound), is displaced by another drug, such as aspirin, potentiating warfarin’s potent anticoagulant effects. Of note in dental prescribing, similar interaction at protein-binding sites occurs with warfarin and most nonsteroidal anti-inflammatory drugs (NSAIDs). Significant increased bleeding risk warrants careful review of the international normalized ratio values, as well as physician consultation when NSAID use is unavoidable.4
INTERACTIONS OF BIOTRANSFORMATION
Variability in drug response is widespread and multifactorial, as estimates suggest most major drugs are effective in only 25% to 60% of the population.2 This irregularity encompasses environmental and genetic factors, including variations in the cytochrome P-450 enzymes (CYPs) involved in drug metabolism in the liver and small intestine. The role of this family of enzymes in the metabolism of drugs and endogenous substances — and their influence on drug effectiveness and toxicity — has been well established.7
Several subfamilies of these enzymes, including CYP2B6, CYP2C9, CYP2C19, CYP2D6, and most notably CYP3A4, are involved in oxidative reactions during first-pass metabolism of more than 90% of clinically important drugs.8 Found in most tissues, including the intestine, lung, adrenal gland, brain, gonads, heart, and even skin and nasal/tracheal mucosa, CYPs are most abundant in the liver, comprising about 2% of hepatic microsomal protein.8 Metabolism (or, more accurately, biotransformation) occurs mostly in the liver, and is the main route of elimination of more than 70% of currently used drugs.8 While oxidation of drugs by P450 enzymes most often results in their inactivation, some drugs, termed pro-drugs, become active as a result.7,9 In addition to codeine (Table 2), some examples of pro-drugs are the anticancer drug tamoxifen, antiplatelet drugs clopidogrel, ticagrelor and prasugrel, and the antihistamine loratadine.9
Because multiple drugs, as well as vitamins, herbal supplements and foods, can be metabolized by the same enzyme, interactions — including an increase or decrease in drug concentration —can occur, making these drugs potentially toxic or ineffective. Various substances can function as inducers or inhibitors of CYPs. When a particular CYP is induced by Drug A, for example, more CYPs are produced in response, and more enzymes can metabolize their other substrates faster. This can result in more efficient inactivation and therefore lower plasma concentration and availability of Drug B that is metabolized by the same CYP at its site of action. This will render Drug B ineffective, and if the two drugs have to be taken together, as is often the case, a dose adjustment of Drug B may be required to maintain its effectiveness. Conversely, if Drug A is an inhibitor of a CYP that also metabolizes Drug B, it will slow metabolism of Drug B, possibly raising its concentration to toxic levels.
While both induction and inhibition of CYPs play a role in drug interactions and their effects, induction requires production of more CYPs in response to the effect of their inducers, and it is a relatively longer process. It is also more predictable at the early stages of drug development, and therefore more preventable.8 Conversely, CYP inhibition, which may be reversible or irreversible depending on the substances and mechanisms of inhibition, is the most common cause of harmful drug-drug interactions, including reduced effectiveness (especially with the pro-drugs), or increased toxicity and adverse effects.7–9
A drug’s effect on the CYPs that metabolize it, potential drug interactions, and patient populations who may have CYPs genetic variabilities are included in drug monographs. These should be carefully reviewed to prevent unexpected drug responses and ensure drug effectiveness and safety. Table 2 includes some drugs encountered in dental practice, their metabolizing CYPs, and potential inducers/inhibitors.
As is often the case in drug research, the discovery of grapefruit juice’s effect on drug metabolism and bioavailability was accidental,10 yet it adversely reacts with many drugs, including calcium channel blockers and statins. While the intent of an early experiment was to evaluate the pharmacokinetics of felodipine (a calcium channel blocker) in the presence of ethanol, grapefruit juice was used only to mask the taste of alcohol. Surprisingly, the subjects experienced more significant blood pressure reduction and incidence of orthostatic hypotension. By ruling out the role of ethanol and potential errors, grapefruit juice was eventually identified as the cause.10 Since then, felodipine interactions have been studied in much detail, and the metabolism of other drugs — including statins, antihistamines, benzodiazepines, cyclosporine, and protease inhibitors — was found to be affected by this strong CYP3A4 inhibitor. Naringin, a flavonoid in grapefruit juice, and its in vivo active metabolite naringenin, as well as other flavonoids, can inhibit drug oxidative metabolism.10 Because grapefruit juice inhibits pre-systemic drug biotransformation by CYP3A4 enzymes in the small intestine, patients with an increased number of these intestinal enzymes can be particularly affected. Those with hepatic insufficiency may also be more sensitive to grapefruit juice interactions. Due to a relatively long half-life of 12 hours, a single glass of grapefruit juice can alter the bioavailability of many drugs, resulting in potentially serious drug interactions and adverse effects.2,10
INTERACTIONS OF ELIMINATION
Drugs are most commonly excreted through the kidneys either as a free drug or metabolites into the urine and cleared from the body at a rate proportional to the blood concentration. Impaired renal function due to kidney disease and the normal aging process is associated with reduced drug clearance. More recently, decreased activity of gastrointestinal enzymes and renal transporters has been linked to chronic and acute kidney disease. This also contributes to prolonged drug elimination and increased risk of toxicity and drug-drug interactions, as multiple drugs may compete for the same excretion sites.11 When prescribing medications, the clinical implications of renal disease or insufficiency require careful evaluation of potential drug interactions. Providers should also consider reducing dosages or increasing dosing intervals due to prolonged clearance rates.4 Therefore, agents with a long half-life, such as tetracyclines, should be avoided in patients with impaired or reduced kidney function, including older adults.
Skaar and O’Connor,12 proponents for utilization of the Beers’ criteria for older adults, identified several drugs commonly prescribed by dentists with the greatest potential for serious drug interactions; these include benzodiazepines, macrolide antibiotics and NSAIDs. For patients with chronic renal failure and those on dialysis, the National Kidney Foundation offers professional and patient support regarding prescription and over-the-counter (OTC) medications, an important consideration for a population at high risk for oral diseases and low dental attendance.13,14
PHARMACODYNAMIC INTERACTIONS
Pharmacodynamic drug actions occur at extra- and intracellular receptor sites of target tissues and non-target tissues, exerting varying degrees of expected and unexpected effects. This may include the interaction of two or more drugs competing for the same receptor sites, producing various outcomes.5 Rather than affecting drug concentration (as with pharmacokinetic interactions), the net effect of the agent(s) on physiological functions may be enhanced or reduced by competitive binding or blocking of receptor sites. Although the ability of drugs to increase or decrease each other’s effects most often leads to adverse outcomes, it can also be beneficial and used therapeutically.3,6 An example of a negative outcome is the well-known interaction of alcohol (ethanol) with benzodiazepines (diazepam, chlordiazepoxide) and barbiturates (phenobarbital), which all bind to their respective sites on γ-aminobutyric acid (GABA) inhibitory GABAA receptors, enhancing GABAergic inhibition and leading to potentially dangerous central nervous system depression.15 Conversely, competitive binding of an opioid antagonist naloxone to the μ and κ opiate receptors reverses the effects of an opioid overdose; this is an example of beneficial pharmacodynamic antagonism.3
Interactions can also occur between drugs that bind to different receptors (noncompetitive), but produce similar effects and thus enhance their actions and potential adverse outcomes.3,5,6 Ethanol binding to the inhibitory GABAA and glycine receptors in the brain, and opioids binding to their opiate receptors occupying different brain regions, produce similar effects and enhance central nervous system depression.3,15
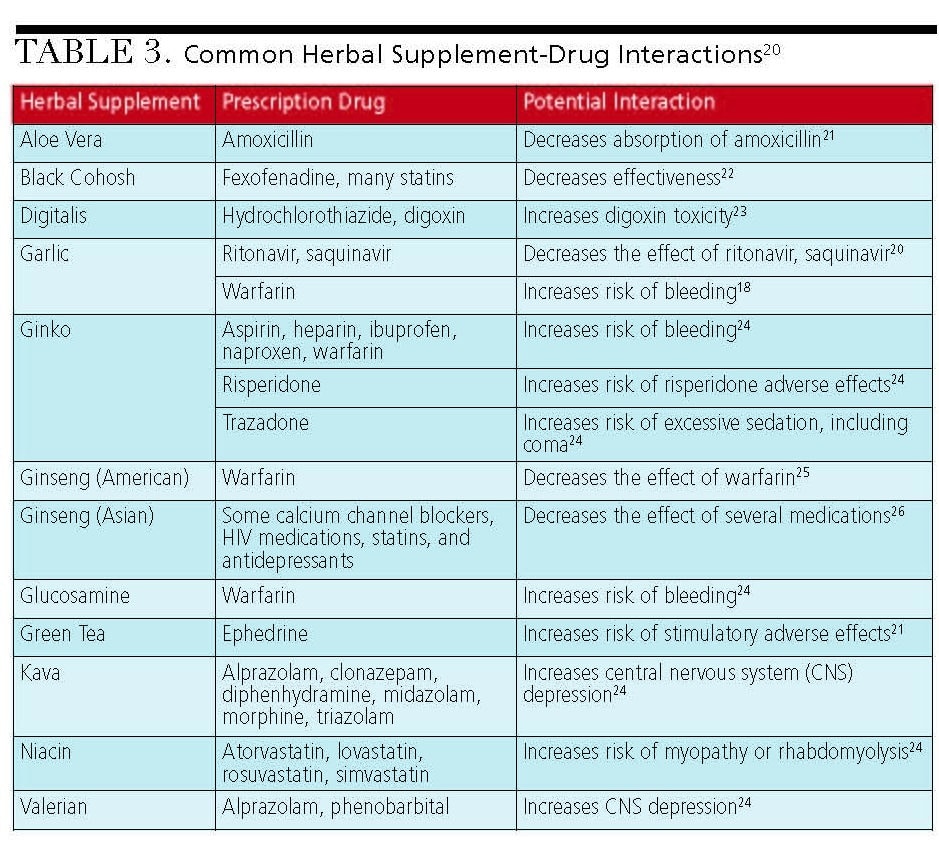
HERBAL SUPPLEMENT-DRUG INTERACTIONS
With nearly 20% to 25% of adults taking an herbal supplement in addition to their prescription medications, it is critical to understand possible drug interactions.16 Despite widespread use of supplements, most patients are ill-informed about potential adverse interactions. Patients are also less likely to list herbal supplements on their medical history, with one study estimating a disclosure rate of 30%.17 Further complicating science’s understanding is the variability in the quality and composition of herbal products, as well as the relative lack of research into the numerous herb-drug combinations.
The main clinical interactions between herbal supplements and prescription drugs involve overlap between their pharmacokinetics and pharmacodynamics. The majority of herb-drug interactions involve pharmacokinetics in which herbal supplements often compete with prescription drugs during ADME pathways. These supplements can act as inducers or inhibitors of various CYPs, resulting in a net decrease or increase in blood concentration of the prescribed medication.2,8 The more overlap between ADME pathways, the more likely a prescribed drug’s concentration will be altered at the site of action. Herb-drug interactions involving pharmacokinetic interactions can often be safely mitigated by altering the dosage of the prescribed medication.16
Although the majority of herb-drug interactions involve pharmacokinetic interactions, some that affect pharmacodynamics will directly alter the prescribed medication’s mechanism of action without altering the blood concentration of the drug. Unlike pharmacokinetic interactions, changing the dosage of the prescribed medication will not counter the effects because the interaction directly affects the mechanism of action, and not the blood concentration of the medication.16
Perhaps the most significant herbal supplement-drug interaction studied involves St. John’s wort. A systematic review of clinical studies in humans has shown that St. John’s wort, often taken to manage depression, is a potent inducer of CYP3A4, CYP2C9, CYP1A2, and the transport protein P-glycoprotein.2 These interactions have been shown to reduce the blood concentrations of warfarin,18 cyclosporine, digoxin, HIV protease inhibitors, selective serotonin re-uptake inhibitors, triptans and oral contraceptives. St. John’s wort is considered a high-risk herbal supplement and patients should be advised to avoid taking it with OTC and prescription medications.19
In light of the potential for adverse drug interactions, oral health professionals should carefully review patients’ use of herbal supplements and examine potential conflicts with prescription and OTC medications, especially when prescribed in the dental office.
CONCLUSION
Drug interactions with other drugs, as well as food, vitamins, minerals and herbal supplements, can occur at any stage of their pharmacokinetics and at their sites of action. Due to the complex underlying chemical and physiological mechanisms, these interactions can result in increased effect and toxicity — or, on the other hand, decreased effect and even inactivation and loss of clinical effectiveness. While some interactions, especially those based on CYPs variability, are idiosyncratic and challenging to predict, many are known and therefore preventable. Oral health professionals should carefully review patients’ medical and drug histories, paying particular attention to prescribed and OTC agents and herbal supplements. This is especially important when new medications are prescribed or recommended as part of dental treatment to ensure the safety and effectiveness of all drugs taken by the patient.
REFERENCES
- U.S. Food and Drug Administration. Preventable Adverse Drug Reactions: A Focus on Drug Interactions. Available at: https://www.fda.gov/drugs/drug-interactions-labeling/preventable-adverse-drug-reactions-focus-drug-interactions. Accessed February 3, 2020.
- Wilkinson GR. Drug metabolism and variability among patients in drug response. N Engl J Med. 2005;352:2211–2221.
- Hersh EV, Moore PA. Adverse drug interactions in dentistry. Periodontol 2000. 2008;46:109–142.
- Dawoud BES, Roberts A, Yates JM. Drug interactions in general dental practice — considerations for the dental practitioner. Br Dent J. 2014;216:15–23.
- Brunton LL, Chabner BA, Knollmann, Björn C. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York: McGraw Hill; 2011.
- Bushra R, Aslam N, Khan AY. Food-drug interactions. Oman Med J. 2011;26:77–83.
- Metabolism of Drugs and Other Xenobiotics. Hoboken, N.J: John Wiley & Sons, Ltd; 2012.
- Pelkonen O, Turpeinen M, Hakkola J, Honkakoski P, Hukkanen J, Raunio H. Inhibition and induction of human cytochrome P450 enzymes: current status. Arch Toxicol. 2008;82:667–715.
- Ortiz de Montellano PR. Cytochrome P450-activated prodrugs. Future Med Chem. 2013;5:213–228.
- Bailey DG, Malcolm J, Arnold O, Spence JD. Grapefruit juice-drug interactions. Br J Clin Pharmacol. 1998;46:101–110.
- 11. Miners JO, Yang X, Knights KM, Zhang L. The Role of the kidney in drug elimination: Transport, metabolism, and the impact of kidney disease on drug clearance. Clin Pharmacol Ther. 2017;102:436–449.
- Skaar DD, O’Connor H. Potentially serious drug-drug interactions among community-dwelling older adult dental patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;112:153–160.
- Costantinides F, Castronovo G, Vettori E, et al. Dental care for patients with end-stage renal disease and undergoing hemodialysis. Int J Dent. 2018;2018:1–8.
- National Kidney Foundation. Available at: https://www.kidney.org/. Accessed February 3, 2020.
- Lobo IA, Harris RA. GABAA receptors and alcohol. Pharmacol Biochem Behav. 2008;90:90–94.
- Asher GN, Corbett AH, Hawke RL. Common herbal dietary supplement — Drug interactions. Am Fam Physician. 2017;96:101–107.
- Mehta DH, Gardiner PM, Phillips RS, McCarthy EP. Herbal and dietary supplement disclosure to health care providers by individuals with chronic conditions. J Altern Complement Med. 2008;14:1263–1269.
- Spolarich AE, Andrews L. An examination of the bleeding complications associated with herbal supplements, antiplatelet and anticoagulant medications. J Dent Hyg. 2007;81:67–67.
- Henderson L, Yue QY, Bergquist C, Gerden B, Arlett P. St John’s wort (Hypericum perforatum): drug interactions and clinical outcomes. Br J Clin Pharmacol. 2002;54:349–356.
From Decisions in Dentistry. March 2020;6(3):26–28,31.





where do I take the exam? You’re website used to be easier to navigate.
Hi Michael – Please click on ‘purchase course‘ and it will take you to the product page for the course where you can purchase. Once you purchase there will be a button to begin the exam and once you complete the exam there will be a button to print your certificate. If you need additional help, please don’t hesitate to email us at info@belmontpublications.com or via our contact page.