
CE Sponsored by Colgate in Partnership With the American Academy of Periodontology — Key Tenets of Periodontal Debridement
The ultimate goal of nonsurgical periodontal therapy is to preserve, improve and maintain the dentition.
This course was published in the October 2017 issue and expires October 2020. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the prevalence and clinical signs of periodontal disease.
- Describe various approaches to nonsurgical periodontal therapy, as well as adjuncts that are designed to improve outcomes
- Explain the physiological processes involved in the treatment of periodontal disease, and when referral to a periodontist is indicated.
INTRODUCTION
Periodontal debridement is a central component of initial periodontal therapy. Despite clinicians’ best efforts, however, some patients will not respond as expected, and their periodontal health will continue to decline. There are many challenges to periodontal debridement, and one of the biggest is that success depends on patient compliance in maintaining healthy habits and good oral hygiene.
This continuing education article, “Key Tenets of Periodontal Debridement,” provides a timely review of the benefits and limitations of nonsurgical periodontal therapy and adjunctive approaches that can help support optimal treatment outcomes.
Colgate-Palmolive Co. is delighted to have provided an unrestricted educational grant to support this educational series in collaboration with the American Academy of Periodontology.
—Matilde Hernandez, DDS, MS, MBA
Scientific Affairs Manager Professional Oral Care
Colgate Oral Pharmaceuticals
FROM THE AMERICAN ACADEMY OF PERIODONTOLOGY
Amid many advancements in periodontal therapy, debridement, along with scaling and root planing, are often key to treating periodontal disease. Although each has its own insurance code — and uses may vary, based on the clinical situation — these procedures can help restore a patient’s periodontal health.
In this insightful continuing education article, educator and American Academy of Periodontology (AAP) member Keerthana Satheesh, DDS, MS, highlights debridement’s effectiveness, limitations and varied approaches. This procedure is one line of defense against advanced infection that is available to the clinical teams whose work is integral to patient wellness and successful treatment outcomes.
The AAP is proud to work with Decisions in Dentistry and Colgate-Palmolive to provide evidence-based information that supports the highest levels of professional care.
—Steven R. Daniel, DDS
President, American Academy of Periodontology
Periodontal disease is a prevalent condition that affects 47.2% of Americans age 30 and older.1 The clinical signs arise from inflammatory and destructive changes in the periodontium. These clinical signs include formation of periodontal pockets, loss of clinical attachment and resorption of the alveolar bone. The etiology is multifactorial and involves a complex interaction between oral biofilm and the host. Biofilms are diverse and highly organized microbial communities composed of a protective extracellular matrix. Bacterial colonization of the periodontal pockets produces virulence factors that can initiate the inflammatory cascade that can directly damage the host tissue, or cause indirect damage through the inflammatory pathways.2 Examples of inflammatory mediators include arachidonic acid metabolites, such as prostaglandin E2, and cytokines, such as Interleukin 1 and tumor necrosis factor-α.3–5
Prevention is the best approach, and supporting patients in developing an effective self-care regimen is critical, as is patient compliance with a recare schedule. Oral health professionals should recommend antimicrobial oral hygiene products, including dentifrice and mouthrinse, as well as oral hygiene aids, to patients based on their individual needs and risk.
Microbial pathogens that grow in biofilms, however, are highly resistant.2 The most effective treatment to control biofilm — a critical step in successfully managing periodontitis — is physical disruption of the bacteria through scaling and root planing.6 Scaling and root planing are the gold standard in nonsurgical periodontal therapy.
A major goal of periodontal therapy is to suppress the extent of the subgingival bacteria and thereby reduce or eliminate the inflammatory periodontal lesion.2,7–11 Scaling and root planing involve supragingival and subgingival instrumentation. While scaling includes removal of plaque, calculus and stains, root planing involves removing cementum or surface dentin that is contaminated with endotoxins and microorganisms.6 Scaling and root planing are also known as periodontal debridement (Figure 1), a term first coined by Smart et al12 to describe the light overlapping strokes used for instrumenting with a sonic or ultrasonic scaler. Other investigators have described the term more broadly as instrumentation — using both hand and mechanical instruments — that is thorough enough to remove toxic substances from the root surface without over-removal of the cementum. The goal for nonsurgical periodontal therapy is to produce a root that is biologically capable of healthy attachment.13–16
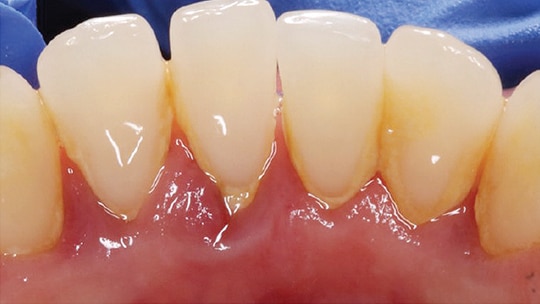
Meticulous instrumentation is a time-consuming and difficult procedure that includes both manual instrumentation and use of sonic or ultrasonic scalers. Compared to sonic and ultrasonic scalers, curets leave a smoother surface.17–19 A drawback of curets, apart from being technique sensitive, is they eventually require sharpening to maintain clinical effectiveness. Numerous studies have also indicated that, in general, manual instrumentation achieves similar clinical end points as the use of sonic or ultrasonic scalers.7,9,10,13,20–22 Current nonsurgical treatment protocol consists of root debridement with sonic or ultrasonic devices and finishing with curets.17,23 Clinical periodontal debridement includes supragingival debridement, subgingival debridement and scaling and root planing. The term is not to be confused with gross debridement.
EFFICACY OF NONSURGICAL THERAPY
Periodontal debridement is effective in decreasing probing depths and bleeding on probing, as well as improving clinical attachment levels.24–27 Greater probing depth reduction is seen in deeper pockets (Table 1). The reduction in probing depths most likely stems from either shrinkage of the gingival margin (which results in a decrease in soft tissue inflammation and edema), or gains in clinical attachment. About half of the probing depth reduction comes from gains in clinical attachment, especially with probing depths of 4 to 6 mm, or 7 mm or more.2,7,28,29 The gain in clinical attachment is mostly from a long junctional epithelium.30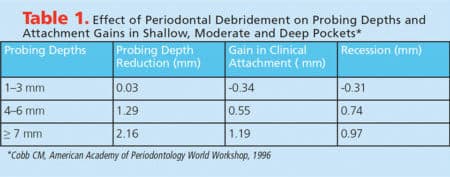
Periodontal debridement changes the microbiological profile. Significant reductions in the percentage of motile microbes and spirochetes,7,31–33 Porphyromonas gingivalis and other gram negative anaerobic microbes31,34–37 were noted following periodontal debridement. A concomitant increase in the number of nonmotile bacteria and percentage of cocci that are associated with periodontal health was also reported.7,31–33,38
Periodontal debridement helps improve glycemic control in patients with diabetes, as research has shown a mean reduction of 0.29% in hemoglobin A1c at three to four months — although ongoing professional periodontal therapy is needed to maintain clinical improvements beyond six months.39 Although the effect of periodontal debridement on decreasing adverse outcomes in pregnancy and childbirth has been debated, a 2012 meta-analysis by Kim et al40 reported a significant decrease in risk of preterm birth with periodontal debridement in high-risk groups. Additional research is needed in this area.
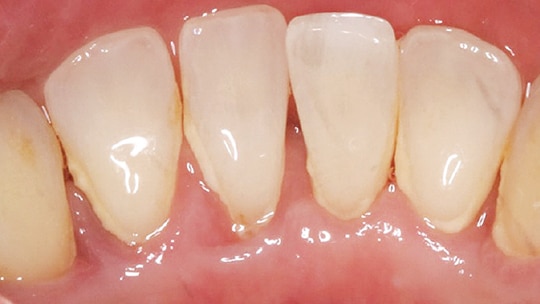
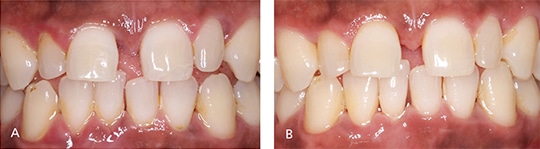
Periodontal debridement must be followed with a reevaluation of treatment outcomes (Figure 2 and Figures 3A and 3B); this is a critical appointment that is scheduled four to six weeks after treatment. Although most healing is complete after six weeks, repair and collagen maturation may continue for an additional six to nine months.2,28,29
LIMITATIONS OF PERIODONTAL DEBRIDEMENT
Several factors have been discussed as causes for less-than-desirable outcomes following periodontal debridement. A patient’s failure to maintain good oral hygiene and noncompliance in returning to regular maintenance care are two common factors.13,41,42 Insufficient debridement may also result in reinfection.7,13,24,36,43 Complete calculus removal is difficult to achieve;44 in one study, for example, complete calculus removal in sites deeper than 5 mm was only possible in 11% of cases.45 The operator’s experience46 and ability to detect calculus,44 distance of the deposit from the cemento-enamel junction,47 and anatomy of the area48,49 are a few possible causes for residual deposits following periodontal debridement.6 In addition, scaling and planing of diseased root surfaces can open up dentinal tubules, allowing invasion by periodontal pathogens into exposed tubules and thereby serving as a reservoir for reinfection of the pocket.2,50,51
Other causes for a limited outcome include systemic disease, such as diabetes, which has a significant impact on long-term success.13,52,53 Genetic susceptibility to periodontitis is also responsible for disease recurrence,13,54,55 while smoking is associated with compromised healing following periodontal debridement.56,57
ADJUNCTIVE APPROACHES
Due the limitations of periodontal debridement, newer tools for improved access have been introduced, as have antimicrobial therapies and adjuncts to conventional instrumentation techniques. Oral health professionals can now choose from a wider and refined selection of power scalers and slim ultrasonic tips, as well as periodontal endoscopes designed to improve visualization. Some operators utilize laser detoxification as adjunctive therapy. Thus far, however, each approach offers only marginal benefits when decontaminating the root surface.24
Adjunctive use of antimicrobials was developed in an effort to improve the clinical and microbiological outcomes of nonsurgical periodontal therapy.58 Every chemotherapeutic adjunct follows a medical mechanical model in which the agent is used to enhance the efficacy of periodontal debridement.59 The antimicrobial adjuncts include application of antiseptics,60,61 or sustained-release local drugs that are designed to prevent plaque accumulation and disinfect the root surfaces and adjacent periodontal tissues.62,63 Systemic approaches include the use of antibiotics and host modulating agents.13,64,65
Locally Delivered Antimicrobials: When used as adjuncts to periodontal debridement, most locally delivered antimicrobials (LDAs) will result in a probing depth reduction of approximately 0.3 to 0.5 mm and attachment level gains of 0.5 to 1 mm.20,66,67 Randomized clinical trials (with a minimum duration of six months) showed clinical attachment gains of 0.4 mm for chlorhexidine chips, 0.64 mm for doxycycline gel, and 0.24 mm for minocycline microspheres.68 Some clinicians recommend the use of LDAs as adjuncts in patients with chronic periodontitis who are at risk for disease progression.59 Local antimicrobials have been shown to provide additional benefits in patients who smoke69 and individuals with periodontitis and diabetes.70
The statement on LDAs from the American Academy of Periodontology (AAP) recommends that clinicians consider their use in chronic periodontitis patients as an adjunct to periodontal debridement when localized recurrent and/or residual 5-mm probing depths with inflammation are still present following conventional therapy. The statement also recommends that alternative treatments should be considered when multiple sites of 5-mm probing depths exist in the same quadrant, the previous use of LDAs has failed to control periodontitis, and in the presence of anatomical defects.71
Systemic Antibiotics: The use of systemic antibiotics has been shown to help improve clinical parameters following periodontal debridement in patients with aggressive periodontitis, refractory periodontitis, and in periodontitis with specific microbiological profiles.72–74 Several antibiotics have been evaluated, among which a combination of amoxicillin and metronidazole was found to be the most effective. Beyond improvements seen with periodontal debridement, the use of amoxicillin and metronidazole resulted in an additional probing depth reduction of 0.58 mm and clinical attachment gains of 0.42 mm on average.24,75 More recently other antibiotics, such as azithromycin, have shown to be valuable adjuncts for patients with deeper pocket probing depths.76
That said, in light of safety concerns and the risk of antimicrobial resistance, the routine use of systemic antibiotics is not recommended. The decision to use these agents should be made carefully for select patients — and only after weighing the risks and benefits. It is critical that antibiotics are not used as stand-alone treatment, and that any prescriptions are accompanied by thorough periodontal debridement.24,74
Host Modulation: Periodontitis is a microbial disease that requires a susceptible host, thus, host modulation is based on the concept of altering the host-mediated inflammatory response. Subantimicrobial-dose doxycycline has been studied extensively as an adjunct to nonsurgical periodontal therapy. In a meta-analysis, a subantimicrobial-dose doxycycline regimen (20 mg twice daily for three months), along with periodontal debridement, was shown to have a significant impact on the clinical parameters at nine months posttreatment.77
Periodontal Endoscopy: This technique utilizes an imaging system, along with the use of specially modified curets and probes, to help clinicians visualize and treat calculus, root surfaces and soft tissue. The ability to view and remove calculus utilizing magnified images is said to improve the clinical results of periodontal therapy. A recent meta-analysis comparing traditional scaling and root planing to periodontal endoscopy reported that the endoscope facilitated superior calculus removal — although the procedure took longer. There was no difference in clinical outcome, however, as measured by the gingival index, bleeding on probing and pocket probing depth.78
Laser Therapy: Laser use in dentistry continues to expand, and various types of lasers have been used as adjuncts for scaling and root planing. Currently, lasers have no defined and accepted protocols for standard usage. The majority of studies involve a small number of subjects and display a significant variation in study design. In addition, results are highly variable between studies. Additional research with larger sample sizes is needed to evaluate the benefit of lasers in periodontal treatment.79 Based on the literature, there is insufficient evidence to suggest that a laser of any given wavelength is superior. The only laser that shows any additional benefit as a periodontal adjunct is the diode laser in photodynamic therapy.79,80
A multidisciplinary panel of the American Dental Association Council of Scientific Affairs concluded the nonsurgical use of lasers other than the diode as adjuncts to scaling and root planing was limited to “expert opinion against” due to uncertainty regarding their clinical benefits and benefit-to-adverse-effects balance.81 A statement from the AAP cautions clinicians to be aware of the potential for root surface damage during in vivo calculus removal with certain lasers. For example, the erbium yttrium aluminum garnet is a hard tissue laser and the operator would not be able to visualize what is being operated upon with the laser.81
Surgical Periodontal Therapy: While there are several therapeutic adjuncts to nonsurgical periodontal therapy, one of the definitive approaches to reducing pocket depths is surgical periodontal therapy. The aim of surgery is to create accessibility for scaling and root planing. A flap access helps operators visualize the root surface during instrumentation. Regeneration is also possible based on the anatomy of the defect. Therefore, when the multiple residual probing depths are present following nonsurgical periodontal therapy, a referral to a periodontist is indicated.
CONCLUSION
The goal of periodontal treatment is to preserve, improve and maintain the dentition. The majority of patients can retain their dentition with appropriate treatment, effective plaque control and regular maintenance. Although periodontal debridement is the cornerstone of nonsurgical periodontal therapy, it is only the first step in treating advanced conditions.
Patients presenting with periodontal disease will need periodontal debridement and subsequent reevaluation to assess the results of the initial therapy. While adjuncts are available to help improve the outcome of debridement in select cases, in advanced cases, the patient may benefit from early referral to the periodontist for surgical treatment and follow-up.
REFERENCES
- Eke PI, Dye BA, Wei L, et al. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91:914–920.
- Cobb CM. Microbes, inflammation, scaling and root planing, and the periodontal condition. J Dent Hyg. 2008;83 (Suppl 3):4–9.
- Kellesarian SV, Malignaggi VR, Abduljabbar T, et al. Efficacy of scaling and root planing with or without adjunct antimicrobial photodynamic therapy on the expression of cytokines in the gingival crevicular fluid of patients with periodontitis: a systematic review. Photodiagnosis Photodyn Ther. 2016;16:76–84.
- Murakami T. Understanding and treatment strategy of the pathogenesis of periodontal disease based on chronic inflammation. Clin Calcium. 2016;26:766–772.
- Ebersole JL, Kirakodu S, Novak MJ, et al. Cytokine gene expression profiles during initiation, progression and resolution of periodontitis. J Clin Periodontol. 2014;41:853–861.
- Deas DE, Moritz AJ, Sagun RS Jr, Gruwell SF, Powell CA. Scaling and root planing vs conservative surgery in the treatment of chronic periodontitis. Periodontol 2000. 2016;71:128–139.
- Cobb CM. Nonsurgical pocket therapy: mechanical. Ann Periodontol. 1996;1:443–490.
- Morrison EC, Ramfjord SP, Hill RW. Short-term effects of initial, nonsurgical periodontal treatment (hygienic phase). J Clin Periodontol. 1980;7:199–211.
- Badersten A, Nilvéus R, Egelberg J. Effect of nonsurgical periodontal therapy. I. Moderately advanced periodontitis. J Clin Periodontol. 1981:8:57–72.
- Badersten A, Nilvéus R, Egelberg J. Effect of nonsurgical periodontal therapy. II. Severely advanced periodontitis. J Clin Periodontol. 1984;11:63–76.
- Pihlstrom BL, Ortiz-Campos C, McHugh RB. A randomized four-year study of periodontal therapy. J Periodontol. 1981;52:227–242.
- Smart GJ, Wilson M, Davies EH, Kieser JB. The assessment of ultrasonic root surface debridement by determination of residual endotoxin levels. J Clin Periodontol. 1990;17:174–178.
- Drisko CH. Nonsurgical periodontal therapy. Periodontol 2000. 2001;25:77–88.
- Checchi L, Pelliccioni GA. Hand versus ultrasonic instrumentation in the removal of endotoxins from root surfaces in vitro. J Periodontol. 1988;59:398–402.
- Cheetham WA, Wilson M, Kieser JB. Root surface debridement — an in vitro assessment. J Clin Periodontol. 1988;15:288–292.
- Drisko CL, Lewis LH. Ultrasonic instruments and antimicrobial agents in supportive periodontal treatment and retreatment of recurrent or refractory periodontitis. Periodontol 2000. 1996;12:90–115.
- Graziani F, Karapetsa D, Alonso B, Herrera D. Nonsurgical and surgical treatment of periodontitis: how many options for one disease? Periodontol 2000. 2017;75:152–188.
- Braun A, Krause F, Frentzen M, Jepsen S. Efficiency of subgingival calculus removal with the vector-system compared to ultrasonic scaling and hand instrumentation in vitro. J Periodontal Res. 2005;40:48–52.
- Kocher T, Rosin M, Langenbeck N, Bernhardt O. Subgingival polishing with a teflon–coated sonic scaler insert in comparison to conventional instruments as assessed on extracted teeth (II). Subgingival roughness. J Clin Periodontol. 2001;28:723–729.
- Kocher T, Rühling A, Momsen H, Plagmann HC. Effectiveness of subgingival instrumentation with power-driven instruments in the hands of experienced and inexperienced operators. A study on manikins. J Clin Periodontol. 1997;24:498–504.
- Yukna RA, Scott JB, Aichelmann-Reidy ME, LeBlanc DM, Mayer ET. Clinical evaluation of the speed and effectiveness of subgingival calculus removal on single-rooted teeth with diamond-coated ultrasonic tips. J Periodontol. 1997; 68:436–442.
- Ioannou I, Dimitriadis N, Papadimitriou K, Sakellari D, Vouros I, Konstantinidis A. Hand instrumentation versus ultrasonic debridement in the treatment of chronic periodontitis: a randomized clinical and microbiological trial. J Clin Periodontol. 2009;36:132–141.
- Ruppert M, Cadosch J, Guindy J, Case D, Zappa U. In vivo ultrasonic debridement forces in bicuspids: a pilot study. J Periodontol. 2002;73:418–422.
- Drisko CL. Periodontal debridement: still the treatment of choice. J Evid Base Dent Pract. 2014;Suppl 33–41:e1.
- Garcia Canas P, Khouly I, Sanz J, Loomer PM. Effectiveness of systemic antimicrobial therapy in combination with scaling and root planing in the treatment of periodontitis: a systematic review. J Am Dent Assoc. 2015;146:150–163.
- Kaldahl WB, Kalkwarf KL, Patil KD. A review of longitudinal studies that compared periodontal therapies. J Periodontol. 1993;64:243–253.
- Mombelli A, Nyman S, Brägger U, Wennström J, Lang NP. Clinical and microbiological changes associated with an altered subgingival environment induced by periodontal pocket reduction. J Clin Periodontol. 1995;22:780–787.
- Greenstein G. Periodontal response to mechanical nonsurgical therapy: a review. J Periodontol. 1992;63:118–130.
- Greenstein G. Nonsurgical periodontal therapy in 2000: a literature review. J Am Dent Assoc. 2000;131:1580–1592.
- Caton JG, Zander HA. The attachment between tooth and gingival tissues after periodic root planing and soft tissue curettage. J Periodontol. 1979;50:462–466.
- Hinrichs JE, Wolff LF, Pihlstrom BL, Schaffer EM, Liljemark WF, Bandt CL. Effects of scaling and root planing on subgingival microbial proportions standardized in terms of their naturally occurring distribution. J Periodontol. 1985;56:187–194.
- Walsh MM, Buchanan SA, Hoover CI, et al. Clinical and microbiologic effects of single-dose metronidazole or scaling and root planing in treatment of adult periodontitis. J Clin Periodontol. 1986;13:151–157.
- Mousquès T, Listgarten MA, Phillips RW. Effect of scaling and root planing on the composition of the human subgingival microbial flora. J Periodont Res. 1980;15:144–151.
- Harper DS, Robinson PJ. Correlation of histometric, microbial, and clinical indicators of periodontal disease status before and after root planing. J Clin Periodontol. 1987;14:190–196.
- Slots J, Mashimo P, Levine MJ, Genco RJ. Periodontal therapy in humans. I. Microbiological and clinical effects of a single course of periodontal scaling and root planing, and of adjunctive tetracycline therapy. J Periodontol. 1979;50:495–509.
- Renvert S, Wikström M, Dahlén G, Slots J, Egelberg J. Effect of root debridement on the elimination of Actinobacillus actinomycetemcomitans and Bacteroides gingivalis from periodontal pockets. J Clin Periodontol. 1990;17:345–350.
- Lowenguth RA, Chin I, Caton JG, et al. Evaluation of periodontal treatments using controlled-release tetracycline fibers: microbiological response. J Periodontol. 1995;66:700–707.
- Singletary MM, Crawford JJ, Simpson DM. Dark-field microscopic monitoring of subgingival bacteria during periodontal therapy. J Periodontol. 1982;53:671–681.
- Simpson TC, Weldon JC, Worthington HV et al. Treatment of periodontal disease for glycaemic control in people with diabetes mellitus. Cochrane Database of Syst Rev. 2015;11:1–153.
- Kim AJ, Lo AJ, Pullin DA, Thornton-Johnson DS, Karimbux NY. Scaling and root planing treatment for periodontitis to reduce preterm birth and low birth weight: a systematic review and meta-analysis of randomized controlled trials. J Periodontol. 2012;83:1508–1519.
- Wilson TG Jr. Supportive periodontal treatment introduction-definition, extent of need, therapeutic objectives, frequency and efficacy. Periodontol 2000. 1996;12:11–15.
- Wilson TG Jr. Compliance and its role in periodontal therapy. Periodontol 2000. 1996;12:16–23.
- Walker CB, Gordon JM, Magnusson I, Clark WB. A role for antibiotics in the treatment of refractory periodontitis. J Periodontol. 1993;64 (Suppl 8):772–781.
- Sherman PR, Hutchens LH Jr, Jewson LG, Moriarty JM, Greco GW, McFall WT Jr. The effectiveness of subgingival scaling and root planing. I. Clinical detection of residual calculus. J Periodontol. 1990;61:3–8.
- Waerhaug J. Healing of the dento-epithelial junction following subgingival plaque control. II: as observed on extracted teeth. J Periodontol. 1978;49:119–134.
- Brayer WK, Mellonig JT, Dunlap RM, Marinak KW, Carson RE. Scaling and root planing effectiveness: the effect of root surface access and operator experience. J Periodontol. 1989;60:67–72.
- Richardson AC, Chadroff B, Bowers GM. The apical location of calculus within the intrabony defect. J Periodontol. 1990;61:118–122.
- Fleischer HC, Mellonig JT, Brayer WK, Gray JL, Barnett JD. Scaling and root planing efficacy in multirooted teeth. J Periodontol. 1989;60:402–409.
- Matia JI, Bissada NF, Maybury JE, Ricchetti P. Efficiency of scaling of the molar furcation area with and without surgical access. Int J Periodontics Restorative Dent. 1986;6:24–35.
- Adriaens PA, De Boever JA, Loesche WJ. Bacterial invasion in root cementum and radicular dentin of periodontally diseased teeth in humans. A reservoir of periodontopathic bacteria. J Periodontol. 1988;59:222–230.
- Giuliana G, Ammatuna P, Pizzo G, Capone F, D’Angelo M. Occurrence of invading bacteria in radicular dentin of periodontally diseased teeth: microbiological findings. J Clin Periodontol. 1997;24:478–485.
- Scannapieco FA. Periodontal disease as a potential risk factor for systemic diseases. J Periodontol. 1998;69:841–850.
- Grossi SG, Genco RJ. Periodontal disease and diabetes mellitus: a two-way relationship. Ann Periodontal. 1998;3:51–61.
- Michalowicz BS. Genetic and inheritable risk factors in periodontal disease. J Periodontol. 1994; 65 (Suppl 5):479–488.
- Engebretson SP, Lamster IB, Herrera-Abreu M, et al. The influence of interleukin gene polymorphism on expression of interleukin-1beta and tumor necrosis factor-alpha in periodontal tissue and gingival crevicular fluid. J Periodontol. 1999;70:567–573.
- Position paper: Tobacco use and the periodontal patient. Research, Science and Therapy Committee of the American Academy of Periodontology. J Periodontol. 1999;70:1419–1427.
- Tonetti MS. Cigarette smoking and periodontal diseases: etiology and management of disease. Ann Periodontol. 1998;3:88–101.
- Herrera D, Matesanz P, Bascones-Martínez A, Sanz M. Local and systemic antimicrobial therapy in periodontics. J Evid Base Dent Pract. 2012;12 (Suppl 3):50–60.
- Paquette DW, Ryan ME, Wilder RS. Locally delivered antimicrobials: clinical evidence and relevance. J Dent Hyg. 2008;83 (Suppl 6):10s–14s.
- Quirynen M, Bollen CM, Vandekerckhove BN, Dekeyser C, Papaioannou W, Eyssen H. Full- vs. partial mouth disinfection in the treatment of periodontal infections: short-term clinical and microbiological observations. J Dent Res. 1995;74:1459–1467.
- Rosling RB, Slots J, Webber RL, Christersson LA, Genco RJ. Microbiological and clinical effects of topical subgingival antimicrobial treatment on human periodontal disease. J Clin Periodontol. 1983;10:487–514.
- Drisko CL, Cobb CM, Killoy WJ, et al. Evaluation of periodontal treatments using controlled-release tetracycline fibers: clinical response. J Periodontol. 1995;66:692–699.
- Jeffcoat M, Bray KS, Ciancio SG, et al. Adjunctive use of a subgingival controlled-release chlorhexidine chip reduces probing depth and improves attachment level compared with scaling and root planing alone. J Periodontol. 1998;69:989–997.
- Crout RJ, Lee HM, Schroeder K, et al. The ‘‘cyclic’’ regimen of low-dose doxycycline for adult periodontitis: a preliminary study. J Periodontol. 1996;67:506–514.
- Golub LM, Lee HM, Ryan ME, Giannobile WV, Payne J, Sorsa T. Tetracyclines inhibit connective tissue breakdown by multiple non-antimicrobial mechanisms. Adv Dent Res. 1998;12:12–26.
- Hallmon WW, Rees TD. Local anti-infective therapy: mechanical and physical approaches. A systematic review. Ann Periodontol. 2003;8:99–114.
- Bonito AJ, Lux L, Lohr KN. Impact of local adjuncts to scaling and root planing in periodontal disease therapy: a systematic review. J Periodontol. 2005;76:1227–1236.
- Smiley CJ, Tracy SL, Abt E, et al. Evidence-based clinical practice guideline on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:525–535.
- Chappke IL, Van der Weijden F, Doerfer C, et al. Primary prevention of periodontitis: managing gingivitis. J Clin Periodontol. 2015;42 (Suppl 16):S71–S76.
- Rovai ES, Souto ML, Ganhito JA, Holzhausen M, Chambrone L, Pannuti CM. Efficacy of local antimicrobials in the nonsurgical treatment of patients with periodontitis and diabetes: a systematic review. J Periodontotol. 2016;87:1406–1417.
- American Academy of Periodontology Statement on Local Delivery of Sustained or Controlled Release Antimicrobials as Adjunctive Therapy in the Treatment of Periodontitis. Available at: joponline.org/doi/ pdf/10.1902/jop.2006.068001. Accessed September 6, 2017.
- Haffajee AD, Socransky SS. Gunsolley JC. Systemic anti-infective therapy. A systematic review. Ann Periodontol. 2003;8:115–181.
- Herrera D, Sanz M, Jepsen S, Needleman I, Roldán S. A systematic review on the effect of systemic antimicrobials as an adjunct to scaling and root planing in periodontitis patients. J Clin Periodontol. 2002;29 (Suppl 3):136–159.
- Herrera D, Alonso B, León R, Roldán S, Sanz M. Antimicrobial therapy in periodontitis: the use of systemic antimicrobials against the subgingival biofilm. J Clin Periodontol. 2008;35 (Suppl 8):45–66.
- Sgolastra F, Gatto R, Petrucci A, Monaco A. Effectiveness of systemic amoxicillin/metronidazole as an adjunctive therapy to scaling and root planing in the treatment of chronic periodontitis: a systematic review and meta-analysis. J Periodontol. 2012;83:1257–1269.
- O’Rourke VJ. Azithromycin as an adjunct to non-surgical periodontal therapy: a systematic review. Aus Dent J. 2017;62:14–22.
- Sgloastra F, Petrucci A, Gatto R, Giannoni M, Monaco A. Long-term efficacy of subantimicrobial-dose doxycycline as an adjunctive treatment to scaling and root planing: a systematic review and meta-analysis. J Periodontol. 2011;82:1570–1581.
- Kuang Y, Hu B, Chen J, Feng G, Song J. Effects of periodontal endoscopy on the treatment of periodontitis: a systematic review and meta-analysis. J Am Dent Assoc. June 18, 2017. Epub ahead of print.
- Cobb CM. Lasers and the treatment of periodontitis: the essence and the noise. Periodontol 2000. 2017;75:205–295.
- Smiley CJ, Tracy SL, Abt E, et al. Evidence-based clinical practice guideline on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:525–535.
- American Academy of Periodontology statement on the efficacy of lasers in the nonsurgical treatment of inflammatory periodontal disease. J Periodontol. 2011;82:513–514.
Featured image by ZLIKOVEC/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. October 2017;3(10):37–42.




