
Asthma’s Effects on Oral Health
An evidence-based asthma action plan will help dental teams support optimal oral health in pediatric patients with asthma.
This course was published in the February 2019 issue and expires February 2022. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Define asthma and discuss its prevalence.
- Identify risk factors for asthma.
- Discuss the effects of asthma medications on oral health.
- List the dental treatment recommendations for this patient population.
An inflammatory disease of the lower respiratory tract, asthma causes the narrowing and inflammation of the airways and hyperresponsiveness of the bronchial airways to various stimuli. This exaggerated reaction results in recurrent symptoms that include wheezing, coughing, dyspnea, increased blood pressure and respiratory rate, excess mucous production, and tightening of the chest.1 This condition can impact the provision of oral health care, so it behooves dental teams to implement and periodically review an asthma action plan designed to support safe and effective care for this patient population.
Asthma ordinarily begins in childhood/adolescence, and — once established — the ongoing inflammatory process leads to underdevelopment of the lungs, further impeding their function.2 More than 6 million U.S. children under the age of 18 have been diagnosed with bronchial asthma.3 Patients with asthma may also experience negative oral health effects, although evidence supporting a casual association is mixed. With the World Health Organization reporting more than 235 million individuals affected by asthma across the globe, oral health professionals should be prepared to treat patients with asthma safely, while also helping them to maintain optimal oral health.4
More than 500 factors can trigger an asthma attack or exacerbate symptoms.5–12 The risk factors for asthma are categorized as either host factors (such as genetics, gender, age, activity status, pregnancy and obesity), or environmental factors (such as allergens, infections, exposure to tobacco smoke, stress, interrupted sleep and pollution).5–13 The genetic predisposition to allergic reactions, or atopy, is the strongest risk factor for developing asthma. Individuals with atopy overproduce immunoglobulin E antibodies and typically present with allergic rhinitis, asthma or eczema.14
Buelo et al15 found that the history of previous asthma attacks, inadequate access to care, poorly managed drug regimens, comorbidity with atopic/allergic diseases, African-American heritage, vitamin D deficiency and poverty have the most significant effect on the risk for asthma attacks among children ages 5 to 12. On the other hand, exposure to tobacco smoke, young age, low parental education and obesity were associated with only a slightly increased risk.15 These results suggest that identifying high-risk children with asthma may help parents/caregivers and health care providers reduce the risk of asthma attacks.
There is no cure for asthma, but symptoms can be controlled through the avoidance or reduction of exposure to asthma triggers and adherence to treatment protocols.16,17 Most patients with asthma are on a medication regimen designed to interrupt the chain reaction that leads to bronchial inflammation. Though these medicines allow patients to lead normal lives, their use is not without risks — some of which present in the oral cavity. As such, oral health professionals should be prepared to effectively treat patients with asthma.
EFFECTS OF ASTHMA MEDICATIONS
The goal of most asthma medications is to directly target the smooth muscle of the respiratory tract with corticosteroids, bronchodilators and anti-inflammatory medications to avert or negate an intense reaction to allergens.17 The goal of asthma therapy is to restore and maintain normal pulmonary function.17
The most common short-term treatment for asthma symptoms is the pulmonary delivery of beta 2 agonists through a rescue inhaler.17,18 These agonists work quickly to relax constricted and inflamed bronchioles at the onset of an asthma attack.17,18 On the other hand, corticosteroids and long-acting beta 2 agonists are taken daily as long-term preventive measures to treat the underlying sustained inflammation that is present even in the absence of allergens.17,18
According to a systematic review by Leung et al,19 the side effects of using inhaled corticosteroids include adrenal insufficiencies, growth suppression and a decrease in bone density. The same review reveals that intermittent administration of slow-acting beta 2 agonists may damage cardiovascular health to a small degree. The pediatric subjects affected exhibited tachycardia, diastolic hypotension and lactic acidosis.19
Regarding oral side effects of asthma medications, some studies demonstrate a higher prevalence of caries among patients with asthma, particularly children, that may be related to the disease and/or its treatment.20 This means asthma may impact dental caries susceptibility either directly through biological changes, such as salivary composition, or indirectly as side effects of the medicines used to control the symptoms of asthma. Prolonged use of beta 2 agonists has been associated with a decrease in salivary production. Thomas et al21 compared patients with asthma who were taking medication to those without asthma and found that salivary secretion of the parotid gland decreased by 36%, while the overall rate of secretion decreased by 26% in patients with asthma. Moreover, obstructed air passageways cause many children and adolescents to breathe through their mouths, further decreasing the presence of moisture in the oral cavity.
Adequate salivary flow is essential to enamel health because it maintains pH balance. Without adequate pH balance, acidic oral conditions ensue and the tooth structure begins to demineralize. For this reason, xerostomia is a major risk factor for caries. However, xerostomia is but one manifestation of asthma medication that impacts enamel soundness.
In addition, beta 2 agonists negatively affect enamel stability in other ways. They work to relax constricted smooth muscles that comprise the airway. This tranquilizing effect occasionally impacts smooth muscles outside of the airway, such as the lower esophageal sphincter.20 In health, the lower esophageal sphincter prevents gastroesophageal acid reflux. However, when relaxed under the influence of beta 2 agonists, the smooth muscle allows erosive gastric acid to enter the mouth.20 Repeated exposure to such acidic conditions poses a significant threat to the dentition — especially the thin enamel and dentin layers in primary teeth — and may result in erosive lesions. Studies show that prolonged use of asthma medications increases the risk of dental erosion.20,22,23 Both aerosol and powdered forms of asthma medications have a pH less than the critical level of 5.5 for enamel dissolution, and come with side effects, such as gastroesophageal reflux and decreased salivary flow.
Asthma medications affect oral health in other ways, as well. The presence of lactose in the inhaler and the fact that many inhalers have a low pH may support the progression of caries. Additionally, oral candidiasis has been associated with the use of inhaled corticosteroids due to an increase in glucose concentration of saliva following inhalation of the corticosteroid carrier vehicle: lactose monohydrate.21 Decreased salivary flow is also thought to contribute to the uninhibited growth of oral candidiasis.21
Research shows an increased presence of calcium in the saliva of children with asthma.20,24 Though the definitive cause of this elevation in calcium levels is unknown, Arafa et al20 speculate the increase in calcium may be attributable to pathologic characteristics of asthma itself or anti-asthmatic bronchodilator drugs. Nevertheless, elevated calcium levels in saliva support the calcification of plaque and deposition of calculus, making optimal oral hygiene all the more necessary.20 An increase in calculus deposits may result in poor gingival and periodontal health. Additionally, diminishing bone density — a potential consequence of the prolonged use of inhaled corticosteroids — can influence the progression of periodontal disease.20
Asthma treatments may also cause acidic pH, dry mouth, anterior open bite, deeper palates, higher frequency of cross bite, atypical swallowing and gingivitis.21,24–27 Studies seeking a correlation between asthma and periodontitis reached varying conclusions. Mehta et al,28 Harrington et al,29 Chakiri et al30 and Arafa et al20 found that children with asthma had increased gingival inflammation compared with children without asthma.
DENTAL CARIES
Robust evidence demonstrates a link between asthma and caries, particularly among children and adolescents.19–21,24,25,31 A systematic review by Agostini et al31 found that individuals with asthma were nearly 1.5 times more likely to develop caries in both primary and permanent dentitions than those without asthma. An earlier systematic review concluded that asthma doubles the risk of caries.32 Yet another study found “a positive correlation between duration of [asthma] and the salivary levels of Streptococcus mutans.”26 Arafa et al20 noted that along with a heightened risk for caries, bronchial asthma is a risk factor for gingival inflammation.
However, the association between caries and asthma is not without controversy. While there are studies demonstrating higher caries prevalence in both dentitions of children with asthma,21,31–34 other studies have failed to show an association between the two chronic conditions.35,36 Maupomé et al37 examined the strength of evidence between 1976 and 2010 and found no causal relationship between asthma and caries. Vasquez et al38 state the impact of asthma on caries in the primary dentition is limited to the nocturnal type. Markovi´c et al39 failed to show a relationship between asthma and caries. This study revealed, however, that children with asthma have poorer oral hygiene than children without asthma, and the amount of corticosteroid inhalant used increased the impact of caries in the primary dentition.39
This discrepancy may be due to limitations within study designs. For instance, previous studies were mostly cross-sectional and consisted of small sample sizes. Additionally, the subjects varied in asthma severity and amounts of medicine consumed. Aside from research design issues, the fact that asthma and caries are chronic and multifactorial diseases makes it difficult to find a relationship between the two.31,35,37,40 There is reason to believe that modifying issues may distort the ability to measure an association between these diseases accurately.31
Varying factors, such as differences in asthma medication regimens, psychological issues, prevalence of mouthbreathing, change in salivary flow rate and properties, low socioeconomic status and ethnicity, increased consumption of sugary drinks, or even the adoption of unhealthy behaviors to pacify patients with asthma may impact study results.21,24,31,35,37–42 For example, McDerra et al43 asserted that parents/caregivers of children with asthma tended to pamper the children with frequent intake of sugary foods and drinks to compensate for the illness.
Treatment Considerations
As part of an asthma action plan, dental teams should create individualized preventive oral health programs according to each child’s risk for oral complications. Table 1 provides an example of an individualized preventive asthma management program.34,41,42,44 This begins with assessment of medicine risks and oral health risk levels. If the patient’s health is stable enough to proceed with treatment, a dental professional must ensure the patient has his or her rescue inhaler on hand and on the bracket table.45,46 During treatment, clinicians should attempt to avoid triggering an asthma attack with aerosols and particles (from dental materials and hard tissue) through proper placement of intraoral trays and suction tips, use of dental dams, and prolonged supine positioning.29,47 The use of nitrous oxide for potentially anxious patients with asthma should be considered because it is neither a respiratory depressant nor an irritant to the bronchial tree.46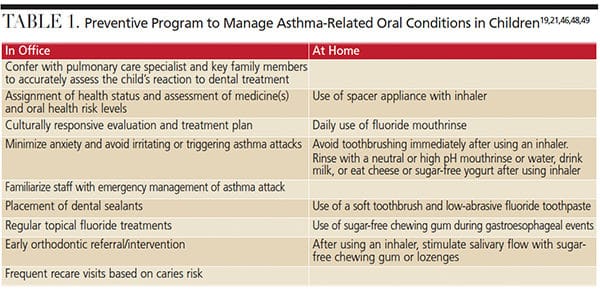
Dental teams are advised to be familiar with emergency management procedures. Children experiencing an acute asthma attack may struggle to breathe and be unable to complete a sentence in one breath. In life-threatening situations, patients may appear cyanotic, have reduced heart and respiratory rates, and exhibit confusion or loss of consciousness.46 If the individual is conscious during an asthma attack, sit the patient upright to take two puffs of his or her inhalant (fast-acting bronchodilator) every 20 minutes for up to an hour.46 If there is no improvement after three doses, administer positive-flow oxygen as supportive treatment and monitor vital signs (oxygen saturation levels should be 93% to 95% in adolescents, and 94% to 98% in children ages 6 to 12).17,46 If there is still no change, contact emergency medical services and administer epinephrine 1:2000 concentration (for a child). If the patient loses consciousness at any time, begin cardiopulmonary resuscitation.29,44,46
When providing oral hygiene instruction, stress the importance of rinsing with neutral or high pH mouthrinses immediately following inhalation of asthma medication, and waiting at least 30 minutes before toothbrushing to allow demineralized enamel to reharden.21,48 Encouraging the development of this simple but highly effective habit may counterbalance the increased caries risk associated with oral inhalation of beta 2 agonists. In order to counteract medication-induced xerostomia, patients can be encouraged to chew sugar-free gum or use an antimicrobial mouthrinse.21
As noted, many inhalers have a low pH and contain sweeteners that enhance their cariogenic potential.20 As such, oral health professionals may recommend the use of a spacer appliance with an inhaler. Use of a spacer device ensures more effective delivery of dry powder inhalers to the lungs, and prevents the medication from scattering to the mouth, throat and stomach.21 A spacer may also assist in reducing the risk of steroid-related oral candidiasis.21
Effective oral self-care, including daily brushing, interdental cleaning, and antimicrobial mouthrinse and fluoride use, is the best defense against caries in any patient population. Both at-home and in-office fluoride applications will aid remineralizing the enamel of a dentition susceptible to acidic environments. Patients at high risk for caries are advised to use a combination of antibacterial and fluoride therapy.49 The prompt placement of dental sealants on erupted permanent molars will also reduce caries prevalence.50
CONCLUSION
Oral health professionals are charged with educating parents/caregivers of children with asthma on the potential oral complications of this chronic condition and to collaborate with medical team members. Undoubtedly, dental teams must take precautions to protect patients’ systemic health, but they also need to help children with asthma manage the possible oral consequences resulting from asthma medication and the biological changes associated with this condition.
REFERENCES
- Bhatt J, Vyas H, Forster D. Management of asthma and allergy. In: Mighten J, ed. Children’s Respiratory Nursing. West Sussex, United Kingdom: John Wiley & Sons Ltd; 2012:113–115.
- Frew AJ. Pathogenesis of asthma. In: Robert R, ed. Clinical Immunology: Principles and Practice. 3rd ed. St. Louis: Mosby, Elsevier; 1997:597–606.
- U.S. Centers for Disease Control and Prevention. National Center for Health Statistics. Available at: https:/ / www.cdc.gov/ nchs/ fastats/ asthma.htm. Accessed January 9, 2019.
- World Health Organization. Asthma: Key Facts. Available at: https:/ / www.who.int/ news-room/ fact-sheets/ detail/ asthma. Accessed January 9, 2019.
- Bateman ED, Hurd SS, Barnes PJ, et al. Global strategy for asthma management and prevention: GINA executive summary. Eur Respir J. 2008;31:143–178.
- Boyce JA, Bochner B, Finkelman FD, Rothenberg ME. Advances in mechanisms of asthma, allergy, and immunology in 2011. J Allergy Clin Immunol. 2012;129:335–341.
- Papadopoulos NG, Christodoulou I, Rohde G, et al. Viruses and bacteria in acute asthma exacerbations — systematic review. Allergy. 2011; 66:458–468.
- Laumbach RJ, Kipen HM. Respiratory health effects of air pollution: update on biomass smoke and traffic pollution. J Allergy Clin Immunol. 2012;129:3–11.
- Khan DA. Exercise-induced bronchoconstriction: burden and prevalence. Allergy Asthma Proc. 2012;33:1–6.
- Brooks SM, Bernstein IL. Irritant-induced airway disorders. Immunol Allergy Clin North Am. 2011:31:747–768.
- Labrecque M. Irritant-induced asthma. Curr Opin Allergy Clin Immunol. 2012:12:140–144.
- Quirce S, Bernstein JA. Old and new causes of occupational asthma. Imunol Allergy Clin North Am. 2011;31:677–698.
- Strunk RC, Sternberg AL, Bacharier LB, Szefler SJ. Nocturnal awakening caused by asthma in children with mild-to-moderate asthma in the Childhood Asthma Management Program. J Allergy Clin Immunol. 2002;110:395–403.
- U.S. Department of Health and Human Services, National Heart, Lung, and Blood Institute. Guidelines for the Diagnosis and Management of Asthma (EPR-3). Available at: nhlbi.nih.gov/ health-topics/ guidelines-for-diagnosis-management-of-asthma. Accessed January 9, 2019.
- Buelo A, McLean S, Julious S. At-risk children with asthma (ARC): a systematic review. Thorax. 2018;0:1–12.
- Chanez P, Bourdin A. Pathophysiology of asthma. In: Castro M, Kraft M, eds. Clinical Asthma. St. Louis: Mosby: 2008.
- Global Initiative for Asthma (GINA): Global strategy for asthma management and prevention. Available at: ginasthma.org. Accessed January 9, 2019.
- Zahran HS, Bailey CM, Damon SA, Garbe PL, Breysse PN. Vital signs: asthma in children — United States, 2001–2016. MMWR Morb Mortal Wkly Rep. 2018;67:149–155.
- Leung JS, Johnson DW, Sperou AJ, et al. A systematic review of adverse drug events associated with administration of common asthma medications in children. PLoS One. 2017;12:e0182738.
- Arafa A, Aldahlawi S, Fathi A. Assessment of the oral health status of asthmatic children. Eur J Dent. 2017;10:357.
- Thomas M, Parolia A, Kundabala M, Vikram M. Asthma and oral health: a review. Aust Dent J. 2010;55:128–133.
- Shaw L, al-Diaigan YH, Smith A. Childhood asthma and dental erosion. ASDC J Dent Child. 2000;67:102–106.
- Al-Diaigan YH, Shaw L, SmitJ AJ. Is there a relationship between ashtma and dental erosion? A case control study. Int J Paediatr Dent. 2002;12:189–200.
- Stensson M. On Oral Health in Young People with Asthma. Available at: https:/ / gupea.ub.gu.se/ handle/ 2077/ 22977. Accessed January 9, 2019.
- Stensson M, Wendt LK, Koch G, Oldaeus G, Lingström P, Birkhed D. Caries prevalence, caries-related factors and plaque pH in adolescents with long-term asthma. Caries Res. 2010;44:540–546.
- Ersin NK, Gulen F, Eronat N, et al. Oral and dental manifestations of young asthmatics related to medication, severity and duration of condition. Pediatr Int. 2006;48:549–554
- Kumar SS, Nandlal B. Effects of Asthma and inhalation corticosteroids on the dental arch morphology in children. J Indian Soc Pedod Prev Dent. 2012;30:242–249.
- Mehta A, Sequeira PS, Sahoo RC, Kaur G. Is bronchial asthma a risk factor for gingival diseases? A control study. N Y State Dent J. 2009;75:44–46.
- Harrington N, Prado N, Barry S. Dental treatment in children with asthma — a review. Br Dent J. 2016;220:299–302.
- Chakiri H, Bahije L, Fawzi R. The effects of the asthma and its treatments on oral health of children: a case control study. Pediatr Dent Care. 2016;1:1–5.
- Agostini BA, Collares KF, dos Santos Costa F, Correa MB, Demarco FF. The role of asthma in caries occurrence — meta-analysis and meta-regression. J Asthma. 2018 Aug 24:1–12.
- Alavaikko S, Jaakkola MS, Tjaderhane L, Jaakkola JJ. Asthma and caries: a systematic review and meta-analysis. Am J Epidemiol. 2011;174:631–641.
- Reddy DK, Gegde AM, Munsgu AK. Dental caries status of children with bronchial asthma. J Clin Pediatr Dent. 2003:27:293–295.
- Shashikiran ND, Reddy VV, Raju PK. Effect of antiasthmatic medication on dental disease: dental caries and periodontal disease. J Indian Soc Pedod Prev Dent. 2007;25:65–68.
- Bahrololoomi Z, Bemanian MH, Ahmadi B. Effect of inhaled medication on dental caries index in asthmatic children. Allergol Immunopathol (Madr). 2018:46:196–200.
- Ehsani S, Moin M, Meihani G, Pourhashemi SJ, Khayatpisheh H, Yarahmadi N. Oral health status in preschool asthmatic children in Iran. Iran J Allergy Asthma Immunol. 2013;12:254–261.
- Maupomé G, Shulman JD, Medina-Solis CE, Ladeinde O. Is there a relationship between asthma and dental caries? A critical review of the literature. J Am Dent Assoc. 2010;141:1061–1074.
- Vasquez EM, Vasquez F, Barrientos MC, et al. Association between asthma and dental caries in the primary dentition of Mexican children. World J Pediatr. 2001:7:344–349.
- Markovi´c D, Peri´c T, Sovti´c A, Mini´c P, Petrovi´c V. Oral health in children with asthma. Srp Arh Celok Lek. 2015;143:539–544.
- Turkistani JM, Farsi N, Almushayt A, et al. Caries experience in asthmatic children: A review of literature. J Clin Pediatr Dent. 2010;35:1–8.
- Satcher D, Nottingham JH. Revisiting Oral Health in America: A report of the surgeon general. Am J Public Health. 2017;107(Suppl 1):S32–S33.
- Zahran HS, Bailey C. Factors associated with asthma prevalence among racial and ethnic groups — United States, 2009–2010 behavioral risk factor surveillance system. J Asthma. 2013;50:583–589.
- McDerra EJ, Pollard MA, Curzon ME. The dental status of asthmatic British school children. Pediatr Dent. 1998;20:281–287.
- Jevon P. Updated posters to help manage medical emergencies in the dental practice. Br Dent J. 2015;219:227–229.
- Yee K, Selwitz-Segal JB. The patient with a respiratory disease. In: Wilkins EM, Wyche CJ, Boyd LD, eds. Clinical Practice of the Dental Hygienist. 12th ed. Philadelphia: Wolters Kluwer; 2017:1121.
- Little JW, Miller CS, Rhodus NL. Dental Management of the Medically Compromised Patient. 9th ed. St. Louis: Elsevier; 2018.
- Khalifa MA, Abouelkheir HM, Khodiar SE, Mohamed GA. Salivary composition and dental caries among children controlled asthmatics. Egyptian Journal of Chest Diseases and Tuberculosis. 2014;63(4):777–788.
- Lussi A, Hellwig E, Ganss C, Jaeggi T. Dental erosion. Oper Dent. 2009;34:251–262.
- Featherstone JDB, Chaffee BW. The evidence for caries management by risk assessment (CAMBRA®). Adv Dent Res. 2018;29:9–14.
- Griffin SO, Barker LK, Wei L, et al. Use of dental care and effective preventive services in preventing tooth decay among U.S. children and adolescents — Medical Expenditure Panel Survey, United States, 2003–2009 and National Health and Nutrition Examination Survey, United States, 2005–2010. MMWR Morb Mortal Wkly Rep. 2014;63(Suppl):54–60.
Featured image by ANANDABGD/E+/GETTY IMAGES PLUS
From Decisions in Dentistry. February 2019;5(2):40—43.




