
Mastering Subgingival Calculus Removal
Discover the latest strategies and tools for effective subgingival calculus removal to prevent periodontitis and ensure optimal oral health.
Essentially mineralized oral biofilm,1,2 calculus is porous, which allows bacteria to thrive on and inside it.3-5 The presence of subgingival calculus is strongly associated with inflammation in the soft tissue pocket wall and is an important factor in the development and progression of periodontitis.1,5,6 Subgingival calculus provides surfaces that are conducive to adherence and growth of biofilms.7 Calculus is a reservoir for endotoxins and microbial antigens;4,8 thus, its incomplete removal leaves a residuum that contributes to reinfection of the periodontal pocket and recurrence of periodontitis.8
A root no longer protected by the periodontal ligament or junctional epithelium is readily colonized by bacteria that may, over time, result in mature biofilm and subgingival calculus. Exposed root surfaces are uneven and hypermineralized; as a result, lacunae, resorptive defects and mounds are common.
Bacterial lipopolysaccharide (endotoxin) penetrates and adsorbs to the roots of periodontally diseased teeth.9 Attachment loss also exposes cementum into which biologically active products can diffuse. Although weakly bound, endotoxin can wreak havoc on healing following inadequate periodontal treatment.10 Bench studies have demonstrated that removal of surface endotoxin via manual or ultrasonic instrumentation or by rinsing are all effective. Unfortunately, SRP alone does not fully eliminate endotoxins. Retoxification of treated surfaces may occur quickly, reinforcing the need to establish brief intervals between periodontal maintenance appointments.
INSTRUMENTATION OF SUBGINGIVAL CALCULUS
Ease of calculus removal is related to location, hardness, composition, mode of attachment, and instrument sharpness — factors that are mediated by biofilm and surface irregularities. Given the difficulties of removing subgingival calculus during SRP, debates regarding optimal instrumentation focus on manual versus ultrasonic and/or sonic instrumentation versus fine-grained, tapered finishing burs.11 A related debate centers on removal of residual cementum and underlying dentin by overly aggressive instrumentation of the root surface. Thus, clinicians often face a conundrum; namely, trying to blindly remove as much subgingival biofilm and calculus as possible seeking to achieve a smooth root surface, but without removing excessive amounts of tooth material. Such challenges led to the development of advanced instruments, such as the periodontal endoscope and videoscope.5,6,12,13 The use of periodontal endoscopes or videoscopes by a skilled clinician can reduce the need for traditional periodontal surgeries.14,15 Figure 1 depicts a view of retained calculus through a periodontal endoscope.
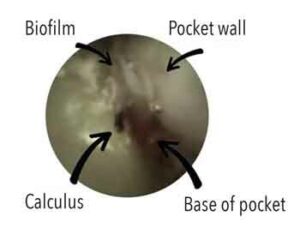
Studies comparing manual to ultrasonic instrumentation with or without surgical flap reflection report no significant difference between the two.11,16 Research also reveals that neither method removes all subgingival biofilm.11,17 The combined use of curets and an ultrasonic scaler appears more effective than either method alone. Studies show that regardless of the type of instruments employed, experienced clinicians can be effective at removing subgingival calculus. Furcations, CEJ sites, interproximal root flutings, deeper pockets, and multirooted teeth are most likely to exhibit residual calculus following SRP. Better results are more likely when manual and ultrasonic instrumentation are combined, or when either is followed by subgingival powder air-polishing.17,18 Although ineffective against calculus, air-polishing using glycine or erythritol powders appears as effective as other approaches for removal of subgingival biofilms.18,19
Lasers are among the most recent additions to the armamentarium. Used either as monotherapies or as adjuncts, lasers have been used to mitigate SRP’s traditional inadequacies in the hope of increasing desired clinical outcomes.20 There is an absence of strong evidence supporting the effectiveness of lasers as adjuncts to SRP.21-23 More high-quality research is needed in this area.
References
- Akcali A, Lang NP. Dental calculus: The calcified biofilm and its role in disease development. Periodontol 2000. 2018;76:109–115.
- Tan BT, Mordan NJ, Embleton J, Pratten J, Galgut PN. Study of bacterial viability within human supragingival dental calculus. J Periodontol. 2004;75:23–29.
- Moolya NN, Thakur S, Ravindra S, Setty SB, Kulkarni R, Hallikeri K. Viability of bacteria in dental calculus — a microbiological study. J Indian Soc Periodontol. 2010;14:222–226.
- Galgut PN, Mordan N, Newman HN. A transmission electron microscopy study of supragingival calculus. J Int Acad Perio. 2001;3:31–37.
- Wilson TG, Harrel SK, Nunn ME, Francis B, Webb K. The relationship between the presence of tooth-borne subgingival deposits and inflammation found with a dental endoscope. J Periodontol. 2008;79:2029–2035.
- Wilson TG, Carnio J, Schenk R, Myers G. Absence of histologic signs of chronic inflammation following closed subgingival scaling and root planing using the dental endoscope: human biopsies — a pilot study. J Periodontol. 2008;79:2036–2041.
- Nyman S, Westfelt E, Sarhed G, Karring T. Role of diseased root cementum in healing following treatment of periodontal disease. A clinical study. J Clin Periodontol. 1988;15:464–468.
- Mandel ID. Calculus update: prevalence, pathogenicity and prevention. J Am Dent Assoc. 1995;126:573–580.
- Hughes FJ, Smales FC. The distribution and quantitation of cementum-bound lipopolysaccharide on periodontally diseased root surfaces of human teeth. Arch Oral Biol. 1990;35:295–299.
- Pitaru S, Soldinger M, Madgar D, Metzger Z. Bacterial endotoxin inhibits migration, attachment, and orientation of human gingival fibroblasts in vitro and delays collagen gel contraction. J Dent Res. 1987;66:1449–1455.
- Oda S, Nitta H, Setoguchi T, Izumi Y, Ishikawa I. Current concepts and advances in manual and power-driven instrumentation. Periodontol 2000. 2004;36:45–58.
- Michaud RM, Schoolfield J, Mellonig JT, Mealey BL. The efficacy of subgingival calculus removal with endoscopy-aided scaling and root planing: a study on multirooted teeth. J Periodontol. 2007;78:2238–2245.
- Kuang Y, Hu B, Chen J, Feng G, Song J. Effects of periodontal endoscopy on the treatment of periodontitis: A systematic review and meta-analysis. J Am Dent Assoc. 2017;148:750–759.
- Harrel SK, Wilson TG Jr, Rivera-Hidalgo F. A videoscope for use in minimally invasive periodontal surgery. J Clin Periodontol. 2013;40:868–874.
- Harrel SK, Nunn ME, Abraham CM, Rivera-Hidalgo F, Shulman JD, Tunnell JC. Videoscope assisted minimally invasive surgery (VMIS): 36-month results. J Periodontol. 2017;88:528–535.
- Drisko CL, Cochran DL, Blieden T, et al. Position paper: sonic and ultrasonic scalers in periodontics. Research, Science and Therapy committee of the American Academy of Periodontology. J Periodontol. 2000;71:1792–1801.
- Cobb CM. Non-surgical pocket therapy: mechanical. Ann Periodontol. 1996;1:443–490.
- Wennstrom JL, Dahlen G, Ramberg P. Subgingival debridement of periodontal pockets by air-polishing in comparison with ultrasonic instrumentation during maintenance therapy. J Clin Periodontol. 2011;38:820–827.
- Flemmig TF, Arushanov D, Daubert D, Rothen M, Mueller G, Leroux BG. Randomized controlled trial assessing efficacy and safety of glycine powder air polishing in moderate-to-deep periodontal pockets. J Periodontol. 2012;83:444–452.
- Ishikawa I, Aoki A, Takasaki AA, Mizutani K, Sasaki KM, Izumi Y. Application of lasers in periodontics: true innovation or myth? Periodontol 2000. 2009:50:90–126.
- Smiley CJ, Tracy SL, Abt E, et al. Systematic review and meta-analysis on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:508–524.
- Chambrone L, Ramos UD, Reynolds MA. AAP best-evidence consensus on infrared laser wavelengths for the treatment of moderate to severe periodontitis. A systematic review and meta-analysis. J Periodontol. 2018;89:743–765.
- Mills MP, Rosen PS, Chambrone L, et al. The efficacy of laser therapy used alone or as an adjunct to non-surgical and surgical treatment of periodontitis and peri-implant diseases: an AAP best-evidence consensus report. J Periodontol. 2018;89:737–742.
This information originally appeared in Rethman MP, Cobb CM, Sottosanti JS, Sheldon LN, Harrel SK. Scaling and root planing remain key to successful periodontal therapy. Decisions in Dentistry. 2021;7(9):25-31.


