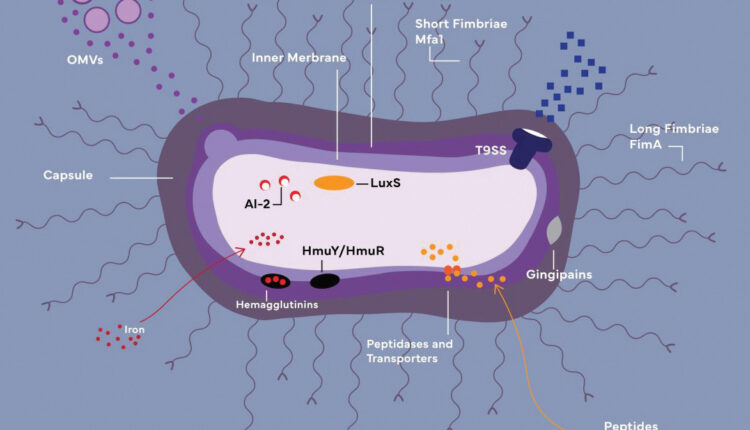
OMVs: Outer membrane vesicles
LPS: Lipopolysaccharide
AI-2/LuxS: Components of quorum-sensing system
HmuY/HmuR: Iron-acquiring transporters (function together with hemagglutinins and gingipains)
Credit: Cheyenne Syrek
Thriving in the Microbiome
Porphyromonas gingivalis gains strength from storing portions of the energy-consuming systems discarded by other microbes.
Through an intricate organization of unique molecular co-aggregation, Porphyromonas gingivalis persists within the host by coupling with other microbes and storing portions of the energy-consuming systems discarded by other microbes, subsequently serving the entire bacterial community’s function and growth. P. gingivalis releases gingipains to mediate interactions between complement receptors and to create a nutrient-rich but oxygen-deficient environment as a protective safeguard from the host immune response, thus permitting co-microbe unions to exist.1–3
The co-infectious microbes bond by exchanging gene-coding signals using quorum sensing — a bacterial communication mechanism that involves cleaving of specific cellular signaling molecules.3 As P. gingivalis co-aggregates with complement species like Treponema denticola and Tannerella forsythia, toxic inflammatory outcomes are averted as they breakdown and immobilize numerous antimicrobial complexes and enzymes discharged by neutrophils.1
The latest research on P. gingivalis co-aggregation with Streptococcus gordonii and Filifactor alocis has garnered much interest because of their co-virulence capabilities. Through a complex adhesion system of short and long fimbria, these specific co-aggregates can influence the overall genotype profile of the microbiome and immune response.1,3 F. alocis, a Gram-positive obligate anaerobe is exceptionally resistant to oxidative stress and can vastly modify the microbiome with its own potent virulence factors.4 The union between P. gingivalis and F. alocis form a strong pathobiont bond increasing proliferation of the junctional epithelium and infiltration of collagen fibers in the gingival epithelial cells.1
Similarly, S. gordonii a well-known opportunistic pathogen, strengthens P. gingivalis virulence with its capacity to permeate the pulp tissue and enter the circulatory system.5 The synergistic action between P. gingivalis and S. gordonii destabilizes the immune response in favor of biofilm growth and intensifies oxidative stress, thus sustaining periodontal inflammatory responses beneficial for the survival and proliferation of P. gingivalis.1,2
Perhaps one of the most intriguing abilities of P. gingivalis is its role as both inhibitor and facilitator of cell death. On one hand, P. gingivalis can influence chronic inflammation by precluding programmed cell death (apoptosis), which can be described as controlled cell death without lysis. This prolongs inflammation by longer host cell survival, followed by their lytic death and release of harmful cellular components into the environment, further exacerbating inflammatory response and providing cellular and DNA fragments as nutrients for P. gingivalis and its pathobionts. Conversely, this bacterium can induce suicide of the host’s and other bacterial cells, termed “fratricide,” once again taking advantage of the released cell fragments as nutrients and further facilitating dysbiosis.1
Before P. gingivalis can survive and populate within a host, it must penetrate the host’s exterior protective barriers and transmute the microbiome to a beneficial state. P. gingivalis accomplishes this mission with the assistance of its very potent virulence factors: capsule, outer membrane proteins and vesicles, lipopolysaccharide (LPS), hemagglutinins, fimbriae, gingipains, and others (Figure 1).6–8
References
- Chopra A, Bhat SG, Sivaraman K. Porphyromonas gingivalis adopts intricate and unique molecular mechanisms to survive and persist within the host: a critical update. J Oral Microbiol. 2020;12:1801090.
- Hajishengallis G, Lamont RJ. Breaking bad: manipulation of the host response by Porphyromonas gingivalis. Eur J Immunol. 2014;44:328–338.
- Gerits E, Verstraeten N, Michiels J. New approaches to combat Porphyromonas gingivalis biofilms. J Oral Microbiol. 2017;9:1300366.
- Aja E, Mangar M, Fletcher HM, Mishra A. Filifactor alocis: recent insights and advances. J Dent Res. 2021;100:790–797.
- Park OJ, Kwon Y, Park C, et al. Streptococcus gordonii: pathogenesis and host response to its cell wall components. Microorganisms. 2020;8:1852.
- Mysak J, Podzimek S, Sommerova P, et al. Porphyromonas gingivalis: major periodontopathic pathogen overview. J Immunol Res. 2014;2014:e476068.
- Mei F, Xie M, Huang X, et al. Porphyromonas gingivalis and its systemic impact: current status. pathogens. 2020;9:944.
- Jia L, Han N, Du J, Guo L, Luo Z, Liu Y. Pathogenesis of important virulence factors of Porphyromonas gingivalis via toll-like receptors. Front Cell Infect Microbiol. 2019;9:262.
This information originally appeared in Marsh I, Matthews A. Porphyromonas gingivalis’ role in the pathogenesis of periodontal disease. Decisions in Dentistry. 2023;9(2):42-45.


