
Managing Hypersensitive Dentin
The following therapies can be used to treat hypersensitivity.
This course was published in the January 2017 issue and expires 01/31/2020.The author has no commercial conflicts of interest to disclose.This 2 credit self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Discuss the incidence of dentinal hypersensitivity, and the patient populations most commonly affected.
- Explain the goals of treating this common condition.
- List various in-office strategies for managing sensitive dentin.
- Identify alternate causes for dentinal hypersensitivity-like hyperesthesia, and dental procedures that can result in sensitivity.
Dentinal hypersensitivity (DH) is a common condition that afflicts 10% to 30% of adults.1 The pain produced by DH can range from subtle (i.e., a minor annoyance) to severe, in which it disrupts daily activities. It can significantly impact patients’ quality of life; this includes the ability to comfortably perform routine oral hygiene. Often, patients may attempt to circumvent sensitivity by modifying habits or behaviors; examples include avoiding hot or cold foods and beverages, and/or eliminating offending items from their diets. Compared to the general population, patients with periodontitis, smokers with periodontitis, and patients with gingival recession tend to have higher incidence of DH. Generally the distribution of DH in the dentition favors maxillary premolars and molars.2 Individuals between 20 to 40 years old present with the greatest incidence of DH; in part, this may be explained by physiologic changes in dentin permeability with aging.3
This article will review the chairside treatment options that can be considered along with counseling and at-home therapy, which remain integral to effective management. Dental professionals should be able to provide recommendations for toothpastes, mouthrinses and pastes to help patients initially address DH at home.4 These strategies are minimally invasive and treat general regions (e.g., multiple teeth or an arch) of exposed cervical dentin, nonspecifically.
Clinical treatments represent an additional level of therapy, and are based on hydrodynamic theory, a well-accepted understanding of sensitivity’s mechanism of action5 in which pain results from fluids within exposed dentinal tubules being disturbed by either temperature, physical or osmotic changes (Figure 1). These fluid movements stimulate a baroreceptor that produces a neural signal. For example, when thermal stimulus is applied to a cervical site with patent dentinal tubules it causes fluid movement within the tubules, resulting in depolarization of nociceptors. Similarly, applying air will desiccate the surface, prompting peripheral flow of fluid toward the dehydrated surface (Figure 2). This fluid movement also depolarizes the nociceptors that evoke the same response. Consequently, the treatment of DH — whether in-office or at home — focuses on two goals: occluding the dentinal tubules, and otherwise impeding the stimulation of pulpal nociceptors.
In-office management can be delineated into four principal approaches. Chemical occlusion of tubules is the main strategy employed with the use of fluoride applications, gluteraldehyde agents, oxalates and calcium-containing agents. Physical blocking of the dentinal tubules results from strategies utilizing application of restorative materials, such as resins, bonding agents and glass ionomers. Nerve desensitization is an approach used in a limited number of chairside products and clinical studies involving the active agent potassium nitrate, a component of many over-the-counter sensitivity dentifrices. Lasers represent perhaps the most contemporary strategy in chairside management of cervical dentinal hypersensitivity. Furthermore, a number of recent studies evaluate the use of lasers in combination with the other aforementioned agents.
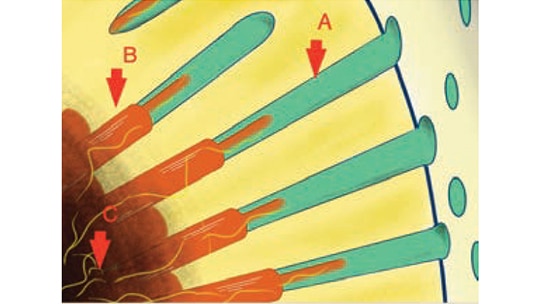
predispose a site to dentinal hypersensitivity. When the smear layer is removed, open
dentin tubules are exposed. In this image, A represents a dentinal tubule; B depicts an
odontoblast and the extending process with proximally associated nerves (yellow); and
C shows the nerves in the pulpal region. Temperature, mechanical, evaporative and
chemical stimuli can change the rate of fluid flow in the tubule, which fires the branches
of the trigeminal sensory nerves (yellow).
FLUORIDE THERAPY
In-office fluoride applications include gels or varnishes applied to the affected areas. These produce a partial or complete blockage of dentinal tubules by forming calcium fluoride or calcium-phosphate precipitates. In dentifrices, fluoride is found in combination with other tubule-obstructing agents that contain potassium or oxalates. When compared with a baseline, a randomized clinical study found two chairside varnishes to be equally effective in reducing dentinal symptomology.6 These 5% sodium fluoride varnishes produced sustained improvements for up to six months. A sodium fluoride gel (1.23%) and stannous fluoride (0.63% in an aqueous solution) also showed promise.7,8 Many products are available in a convenient singleuse/ dose format. In addition, topical fluorides are a common material already found in many dental practices.
GLUTARALDEHYDE/HYDROXYETHYL METHACRYLATE
Products containing 5% glutaraldehyde and 35% hydroxyethyl methacrylate (HEMA) are also used to treat DH. Kakaboura et al9 demonstrated that use of these products can alleviate symptoms by 5% to 27% for seven to nine months. Glutaraldehyde/HEMA reacts with serum albumin proteins in dentinal fluid to form a precipitate that narrows or fully blocks dentinal tubules. Scanning electron microscopy and confocal laser scanning microscopy also show that glutaraldehyde/HEMA causes serum protein coagulation (i.e., denaturation) that further occludes tubules.10
OXALATES
Oxalates form a precipitate that occludes patent dentinal tubules to help relieve sensitivity. Based on a systematic review of available controlled clinical trials, one variant of this compound, 3% monohydrogen-monopotassium oxalate, demonstrated a slight beneficial effect over a placebo.11 As with other attempts at synthesis of the clinical literature, however, there were notable challenges due to the heterogeneity and lack of standardization of pain measurement and DH diagnostic criteria. Symptomology is reduced with a combination strategy in which an oxalic acid desensitizing agent is applied prior to composite resin restoration. This randomized controlled clinical trial treated patients with noncarious cervical lesions and hypersensitivity, and produced reduced sensitivity at six months posttreatment.12
ARGININE-CALCIUM CARBONATE
Shown to be clinically effective, a topical paste containing 8% argininecalcium carbonate inhibits the hydrodynamic mechanism of DH by plugging patent tubules.13 The mechanism of this agent relies on the natural biological formation of a protective surface layer consisting of a precipitate of salivary glycoproteins with calcium phosphate. In a study of 68 patients, a single prophylaxis-cup application of arginine-calcium carbonate paste provided statistically significant reductions in DH immediately, with results lasting about a month.14 This product may help manage post-instrumentation discomfort in patients with a history of DH.
ADHESIVES AND RESTORATIVE MATERIALS
Bonding agents serve to physically occlude dentinal tubules. Two randomized controlled trials found that adhesives significantly relieved DH for up to a month.15,16 A double-blind, randomized, parallel prospective study compared the effectiveness of a glutaraldehyde/HEMA desensitizer, dentin desensitizer/adhesive combination, and a self-etching adhesive in the chairside treatment of DH. All products helped reduce DH compared with controls, but the glutaraldehyde/HEMA desensitizer and dentin desensitizer/ adhesive combination demonstrated greater reductions than a self-etching adhesive.17 Advances in this area include the use of a light cure over a resin-based adhesive to inhibit tubule fluid flow for longer periods. Other restorative materials, such as composite resin and glass ionomer, may be utilized in scenarios in which a pattern of missing cervical tooth structure exists concomitantly with DH symptomology.
LASERS
Research findings vary regarding the effectiveness of lasers in the clinical treatment of DH. The body of evidence supporting their use, however, is growing. Low-output lasers, such as helium-neon and diode types, may function by interfering with nerve transmission. A systematic review and meta-analysis reported heterogeneity of results, with success rates ranging from 5% to 100%.18 The combination of a low-output laser and glutaraldehyde/HEMA application can improve DH symptoms for up to six months.19
High-output lasers, such as Nd:YAG and CO2 devices, have also been evaluated in clinical studies. The Nd:YAG laser is thought to contract dentinal tubules through the energy transferred by the laser, thereby reducing DH symptoms. Longer follow-up of CO2 laser-treated teeth in vivo at four to six months confirm (via scanning electron microscopy) continued closure of dentinal tubules. High-output lasers may prove effective due to resultant thermal coagulation of proteins in dentinal fluid.20 According to a systematic review,21 the performance of lasers for relieving DH symptoms varies, but new studies report encouraging results.
Additional well-designed clinical trials will help better demonstrate the value of lasers in managing DH. For example, the duration in which DH relief can be achieved using laser irradiation is not conclusive, as long-term recurrence rates are unclear in the published literature. Furthermore, recent studies demonstrate efficacy of lasers in conjunction with a desensitizing agent in managing DH. Compared to varnish or lasers alone, Suri et al22 report the most ideal results in subjects receiving 5% sodium fluoride varnish, followed by irradiation with a 980-nanometer diode laser. The combination of Nd:YAG laser and 5% sodium fluoride varnish appears to derive its efficacy by associated occlusion of patent tubules, as confirmed by scanning electron microscopy.23 The effect of lasers and sodium fluoride varnish is thought to relate to greater adhesion of sodium fluoride gel following laser treatment. In another investigation, the use of a laser combined with arginine-calcium carbonate paste did not further improve efficacy in treating DH. A randomized split-mouth study investigated 8% arginine-calcium carbonate and diode laser application.24 No synergistic effects were demonstrated in the treatment arms, as all except the placebo showed reduced pain response at three months. The merit of combined therapy may hinge upon the precise pairings of laser and desensitizing agent. Toward this end, continued clinical research — with longer prospective longitudinal studies in larger sample sizes — is necessary.
OTHER THERAPIES
Although potassium nitrate is a common ingredient in over-the-counter dentifrices, it is also available in at-home and in-office gels. Potassium nitrate increases extracellular potassium ion concentration, depolarizing the nerve and inhibiting it from repolarizing. The literature regarding its efficacy is mixed, however.25 Strontium salts work in the same manner as potassium nitrate by aiming to desensitize the nerve. Its secondary mechanism involves blocking patent dentinal tubules.26
Calcium phosphate therapies — including amorphous calcium phosphate (ACP), casein phosphopeptide-ACP, bioactive glass and tri-calcium phosphate — are also used to address this condition. By providing additional calcium and phosphate ions to support remineralization of tooth structure, ACP, casein phosphopeptide-ACP, and tri-calcium phosphate may help relieve DH. Bioactive glass creates a hydroxycarbonate apatite layer to block tubules, thus decreasing sensitivity. These technologies are available as in-office gels, varnishes, prophylaxis pastes, professionally dispensed cream and gum, and prescription toothpastes. More in vivo research is needed to demonstrate their effectiveness in DH treatment.
SURGICAL APPROACHES
The coverage of exposed roots using periodontal mucogingival surgery represents another option to address dentinal hypersensitivity. The main rationale for pursuing soft tissue coverage of localized gingival recession is esthetic in nature.27 Unsurprisingly, the vast majority of clinical literature reports treatment outcomes with the “bottom line” focused on quantitative root coverage endpoints. Thus, there is insufficient clinical evidence to conclude that surgical root coverage predictably addresses cervical dentinal hypersensitivity.28 A related conclusion was summarized in clinical guidelines of the American Academy of Periodontology Regeneration Workshop consensus report on soft tissue root coverage procedures. While predictable root coverage is achievable in single-site and multiple-site recessions, especially with traditional subepithelial connective tissue graft procedures, there is a dearth of patient-centered data on effectiveness in managing cervical dentinal hypersensitivity.29,30
PREVENTING HYPERSENSITIVITY
Dental professionals should attempt to eliminate competing diagnoses that might account for a patient’s DH signs and symptomology. Alternate causes for DH-like hyperesthesia include caries, fractured/chipped teeth and faulty restorations. Clinicians must likewise consider recent dental treatments, such as restorative procedures or scaling and root planing, that can induce sensitivity. In order to reduce the risk of DH, care should be taken to avoid over-instrumenting exposed dentin with hand, ultrasonic or polishing modalities.
Factors that can contribute to the development of DH should also be addressed; for example, clinicians can provide oral hygiene instruction to counter overzealous brushing, or counseling to help manage erosion caused by acidic foods. It is important to note that consumption of wine, acidic juices or soft drinks can facilitate the dissolution of dentinal smear layers, thus uncovering previously occluded dentinal tubules. When assessment fails to lead to the cause, patients can be asked to keep symptom diaries to help isolate specific etiologies. Prior to the clinical treatment of DH, a short-term approach may include use of at-home prescription fluoride products and/or calcium phosphate therapies. With these initial strategies exhausted, the second line of management may now involve in-office treatments to defined or targeted cervical regions that experience DH symptoms.
The current body of published clinical research does not offer overwhelming support for one treatment modality over another. In light of this, developing a comprehensive approach that uses at-home and in-office strategies based on the best-available evidence is critical to effectively addressing DH in all patient populations. In this manner, dental professionals can serve patients by providing the means to overcome the discomfort associated with this common condition.
REFERENCES
- Gillam DG. Management of dentin hypersensitivity. Clin Oral Investig. 2015;2:87–94.
- Rees JS, Addy M. A cross-sectional study of dentine hypersensitivity. J Clin Periodontol. 2002;29:997–1003.
- Flynn J, Galloway R, Orchardson R. The incidence of hypersensitive teeth in the West of Scotland. J Dent. 1985;13:230–236.
- Shiau HJ. Dentin hypersensitivity. J Evid Based Dent Pract. 2012;12(Suppl 3):220–228.
- Brannstrom M. The surface of sensitive dentine. An experimental study using replication. Odontol Revy. 1965;16:293–299.
- Ritter AV, de L Dias W, Miguez P, Caplan DJ, Swift EJ Jr. Treating cervical dentin hypersensitivity with fluoride varnish: A randomized clinical study. J Am Dent Assoc. 2006;137:1013–1020.
- Miller S, Truong T, Heu R, Stranick M, Bouchard D, Gaffar A. Recent advances in stannous fluoride technology: Antibacterial efficacy and mechanism of action towards hypersensitivity. Int Dent J. 1994;44(Suppl 1):83–98.
- Armenio RV, Fitarelli F, Armenio MF, Demarco FF, Reis A, Loguercio AD. The effect of fluoride gel use on bleaching sensitivity: A double-blind randomized controlled clinical trial. J Am Dent Assoc. 2008;139:592–597.
- Kakaboura A, Rahiotis C, Thomaidis S, Doukoudakis S. Clinical effectiveness of two agents on the treatment of tooth cervical hypersensitivity. Am J Dent. 2005;18:291–295.
- Ishihata H, Finger WJ, Kanehira M, Shimauchi H, Komatsu M. In vitro dentin permeability after application of Gluma desensitizer as aqueous solution or aqueous fumed silica dispersion. J Appl Oral Sci. 2011;19:147–153.
- Cunha-Cruz J, Stout JR, Heaton LJ, Wataha JC, Northwest PRECEDENT. Dentin hypersensitivity and oxalates: a systematic review. J Dent Res. 2011;90:304–310.
- Corral C, Grez PV, Letelier M, Dos Campos EA, Dourado AL, Fernández GE. Effect of oxalic acid-based desensitizing agent on cervical restorations on hypersensitive teeth: A triple-blind randomized controlled clinical trial. J Oral Facial Pain Headache. 2016;30:330–337.
- Petrou I, Heu R, Stranick M, et al. A breakthrough therapy for dentin hypersensitivity: How dental products containing 8% arginine and calcium carbonate work to deliver effective relief of sensitive teeth. J Clin Dent. 2009;20:23–31.
- Schiff T, Delgado E, Zhang YP, Cummins D, DeVizio W, Mateo LR. Clinical evaluation of the efficacy of an in-office desensitizing paste containing 8% arginine and calcium carbonate in providing instant and lasting relief of dentin hypersensitivity. Am J Dent. 2009;22:8A–15A.
- Ozen T, Orhan K, Avsever H, Tunca YM, Ulker AE, Akyol M. Dentin hypersensitivity: A randomized clinical comparison of three different agents in a short-term treatment period. Oper Dent. 2009;34:392–398.
- Yu X, Liang B, Jin X, Fu B, Hannig M. Comparative in vivo study on the desensitizing efficacy of dentin desensitizers and one-bottle self-etching adhesives. Oper Dent. 2010;35:279–286.
- Patil SA, Naik BD, Suma R. Evaluation of three different agents for in-office treatment of dentinal hypersensitivity: A controlled clinical study. Indian J Dent Res. 2015;26:38–42.
- Kimura Y, Wilder-Smith P, Yonaga K, Matsumoto K. Treatment of dentine hypersensitivity by lasers: A review. J Clin Periodontol. 2000;27:715–721.
- Lopes AO, Eduardo Cde P, Aranha AC. Clinical evaluation of low-power laser and a desensitizing agent on dentin hypersensitivity. Lasers Med Sci. 2015;30:823–829.
- Moritz A, Schoop U, Goharkhay K, et al. Long-term effects of CO2 laser irradiation on treatment of hypersensitive dental necks: results of an in vivo study. J Clin Laser Med Surg. 1998;16:211–215.
- Sgolastra F, Petrucci A, Severino M, Gatto R, Monaco A. Lasers for the treatment of dentin hypersensitivity: A meta-analysis. J Dent Res. 2013;92:492–499.
- Suri I, Singh P, Shakir QJ, Shetty A, Bapat R, Thakur R. A comparative evaluation to assess the efficacy of 5% sodium fluoride varnish and diode laser and their combined application in the treatment of dentin hypersensitivity. J Indian Soc Periodontol. 2016;20:307–314.
- Kumar NG, Mehta DS. Short-term assessment of the Nd:YAG laser with and without sodium fluoride varnish in the treatment of dentin hypersensitivity — a clinical and scanning electron microscopy study. J Periodontol. 2005;76:1140–1147.
- Bal MV, Keskiner I, Sezer U, Açıkel C, Saygun I. Comparison of low level laser and arginine-calcium carbonate alone or combination in the treatment of dentin hypersensitivity: a randomized split-mouth clinical study. Photomed Laser Surg. 2015;33:200–205.
- Addy M. Dentine hypersensitivity: new perspectives on an old problem. Int Dent J. 2002;52:367–375.
- Pashley D. Dynamics of the pulpo-dentin complex. Crit Rev Oral Biol Med. 1996;7:104–133.
- Zaher CA, Hachem J, Puhan MA, Mombelli A. Interest in periodontology and preferences for treatment of localized gingival recessions. J Clin Periodontal. 2005;32:375–382.
- Douglas de Oliveira DW, Oliveira-Ferreira F, Flecha OD, Gonçalves PF. Is surgical root coverage effective for the treatment of cervical dentin hypersensitivity? A systematic review. J Periodontol. 2013;84:295–306.
- Tatakis DN, Chambrone L, Allen EP. Periodontal soft tissue root coverage procedures: A consensus report from the AAP Regeneration Workshop. J Periodontol. 2015;86:S52–S55.
- Oates TW, Robinson M, Gunsolley JC. Surgical therapies for the treatment of gingival recession. A systematic review. Ann Periodontol. 2003;8:303–320
Featured photo by STEVE GSCHMEISSNER/SCIENCE SOURCE
From Decisions in Dentistry. January 2017;3(1):46-49.




