
CE Sponsored by Colgate in Partnership With the American Academy of Periodontology: Managing Dentinal Hypersensitivity in Periodontal Patients
The following strategies can be helpful in preventing, diagnosing and treating dentinal hypersensitivity.
PURCHASE COURSE
This course was published in the May 2017 issue and expires May 2020. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Discuss possible causes and management strategies for dentinal hypersensitivity.
- Explain the prevalence of dentinal hypersensitivity in the general population.
- List the agents and methods of action by which they help reduce sensitivity.
Introduction
Colgate-Palmolive Co. is delighted to have provided an unrestricted educational grant to support this continuing education article — “Managing Dentinal Hypersensitivity in Periodontal Patients” — in collaboration with the American Academy of Periodontology.
Dentinal hypersensitivity can make your patient’s appointment uncomfortable. Recognizing that some procedures may trigger pain in periodontal patients, this article provides a helpful review of technologies available for use today, starting in your office with professionally applied desensitizers or laser therapy, and continuing at home with topical agents, such as dentifrices. We hope you find this article interesting and valuable in clinical practice — and, ultimately, of benefit to your patients.
—Matilde Hernandez, DDS, MS, MBA
Scientific Affairs Manager Professional Oral Care
Colgate Oral Pharmaceuticals
From the American Academy of Periodontology
Dentinal hypersensitivity is both a common symptom of periodontal disease and a side effect of certain periodontal therapies. As such, how can periodontists, general dentists and dental hygienists ensure the comfort and health of patients, both during and after treatment? The solutions may vary, depending on etiology and measures previously taken to reduce or eliminate sensitivity. In this article, educator and American Academy of Periodontology (AAP) member Mia L. Geisinger, DDS, MS, explores the foundations of dentinal hypersensitivity and possible approaches for managing this common, but painful, condition.
Decisions in Dentistry and Colgate-Palmolive Co. to bring you insights that will keep your patients comfortable and well.
—Terrence J. Griffin, DMD
President, American Academy of Periodontology
Dentinal hypersensitivity has been defined as a sharp, painful response to external stimuli applied to exposed dentin.1,2 There are many possible causes that expose dentin and predispose patients to hypersensitivity, including toothbrush abrasion, acid erosion, anatomical factors and gingival recession.3,4 In particular, periodontitis and periodontal therapy can lead to exposure of root surfaces, which may cause sensitivity;5 in fact, its sudden onset can be a major side effect of treatment.6,7 Both surgical and nonsurgical periodontal therapies have been shown to increase the prevalence of hypersensitivity related to gingival recession and/or root denudation.8–10 To ensure optimal care, identification, prevention and treatment strategies to address patient discomfort are critical to allow for effective management of periodontal conditions.
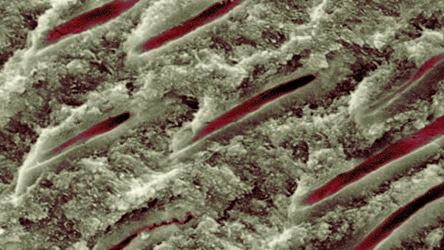
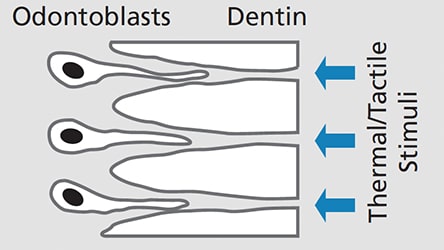
Root sensitivity and dentinal hypersensitivity likely have similar etiology, diagnosis and management.11 While several theories have been proposed to explain hypersensitivity, hydrodynamic theory is the most widely accepted.4,12,13 This holds that sensitivity occurs when tactile, thermal and/or chemical stimuli cause movement of the fluid that surrounds the odontoblastic processes within dentinal tubules (Figure 1). This fluid flow can further stimulate intradental nerves, specifically A-β and A-δ myelinated fibers, causing the short, intense pain associated with this condition.12 Hydrodynamic theory postulates that two conditions must be met on root surfaces for sensitivity to develop: the presence of exposed dentin (“lesion localization”), and open dentinal tubules communicating with a vital pulp that are exposed to stimuli (“lesion initiation”).14,15 Histologic findings support this explanation and demonstrate greater numbers of widened dentinal tubules in sensitive versus nonsensitive teeth (Figure 2).16,17
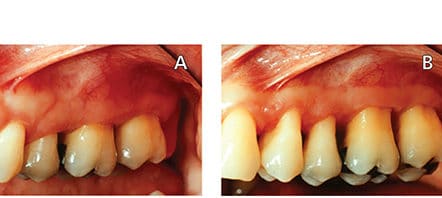
The prevalence of dentinal hypersensitivity in the general population has been reported to range from 4% to 57%,11,18–20 with higher values noted for individuals with periodontitis.18,21 A systematic review also showed that prior to periodontal therapy the prevalence of root sensitivity ranged from 9% to 23%, but it increased to 54% to 55% after treatment.8 Increases in pain intensity after periodontal therapy are generally moderate9 and have been shown to abate approximately four weeks after therapy8,9 — although 10% to 15% of patients experiencing root sensitivity after periodontal treatment may never achieve spontaneous remission of symptoms.22 Root sensitivity has also been reported as long as five years following surgical periodontal treatment.23 Thus, root sensitivity control for periodontal patients is a two-pronged problem: Periodontitis can lead to recession and root exposure as a part of disease progression, and treatment strategies aimed at removing etiologic factors and reducing probing depths lead to further removal of cementum or smear layer and exposure of the dentinal tubules, as well and increased gingival recession (Figures 3A and 3B).
Identification of dentinal hypersensitivity is difficult and may be subjective. For example, in population studies, self-reported rates were significantly higher than those determined by clinical testing,19,20,24–26 indicating a significant component of recall bias and subjectivity. For patients who have recently undergone periodontal therapy, consideration of the timing in postoperative pain onset, as well as the location, severity and reproducibility of the sensitivity symptoms — in combination with the elimination of other possible sources — offer insight into the proper diagnosis.27,28 Some conditions that may elicit similar symptoms include cracked or chipped teeth, fractured or improperly placed metallic restorations, postrestorative sensitivity, caries, hypoplastic enamel, or a congenitally open cemento-enamel junction (CEJ).29 Open CEJs may be a predisposing factor for sensitivity and occur in approximately 10% of the population.30 Care should be taken to rule out alternative causes of tooth pain. The examination should include a thorough medical/dental history and a clinical/radiographic evaluation of the areas presenting with pain (and surrounding areas, if necessary).6
TREATMENT STRATEGIES
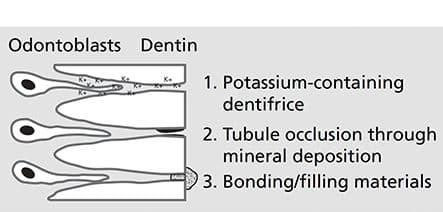
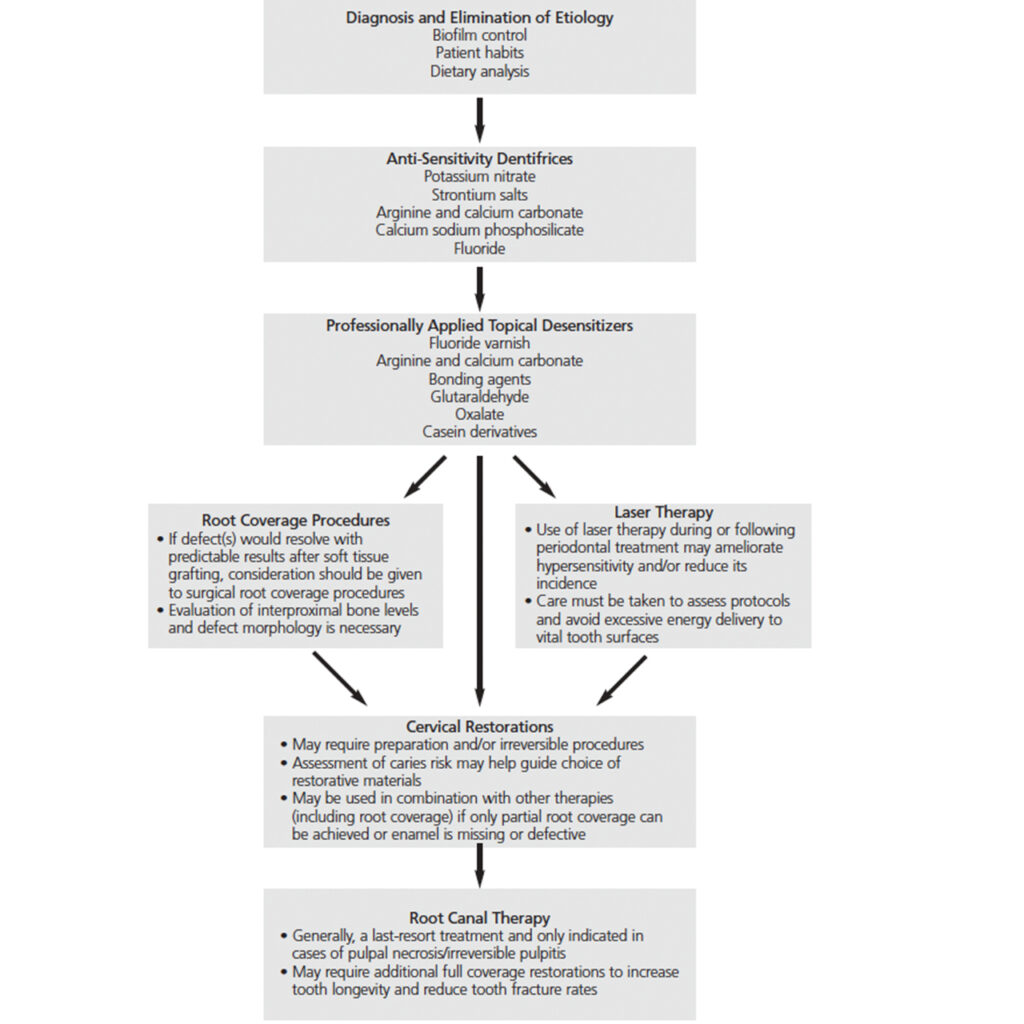
Because the permeability and fluid movement in open, exposed dentinal tubules is the favored theory for stimuli transmission through dentin, treatment principles include reducing or eliminating irritating stimuli, tubule obliteration (i.e., “dentinal plugs”), tubule coverage and preventing transmission of irritants (Figure 4).2,31
Acid exposure from diet and cariogenic biofilm has been related to prevalence and severity of hypersensitivity.14,32,33 While some authors have suggested that mechanical irritation from biofilm control may contribute to sensitivity,4,34 most studies confirm that meticulous biofilm control reduces root sensitivity — both before and after periodontal treatment.9,35–37 In an effort to mitigate the etiologic factors of periodontal disease and minimize root sensitivity following therapy, individualized oral hygiene instruction should be a component of all periodontal care. Biofilm control should be reassessed throughout treatment, including the postsurgical phase. Dietary counseling to limit irritating foods and beverages should also be considered.
TOPICAL AGENTS: DENTIFRICES
Managing dentinal hypersensitivity through agents added to dentifrices is often a first-line therapy and may be combined with in-office treatment. These ingredients — which may include potassium nitrate, strontium acetate, arginine and calcium carbonate, calcium sodium phosphosilicate, and fluoride38 — either reduce nerve conduction or occlude tubules. Many work with differing mechanisms and patients may need to try several products prior to finding relief for their symptoms. They may also need multiple treatment strategies in combination.
Potassium nitrate is believed to depolarize nerves with the tubules and inhibit or reduce their ability to transmit pain signals.39–43 Some studies have demonstrated an improvement in symptoms after compliant use for two weeks in both a 5% or 10% concentration.44–46 Other studies have demonstrated no statistically significant reduction in sensitivity with use.47–49 These variable results may be the result of specific patient populations, differing methods for evaluating dentinal sensitivity, and the difficulty of quantifying pain. The timing of the clinical improvements seen and equivocal data on the relief provided may indicate limited utility for these dentifrices in many patients with sensitivity related to periodontal therapy. However, potassium nitrate has not been shown to induce pulpal changes after use50 and this agent may still offer benefits to some patients.49
Strontium salts’ mechanism of action involves tubule occlusion,51 in which strontium ions exchange with calcium ions, causing the formation of insoluble strontium crystals within exposed tubules, up to an average depth of 5 µm.51–53 Dietary acids have also been shown not to alter occlusion after the precipitation of strontium acetate48 — which may lead to longer-lasting results. Furthermore, clinical studies have demonstrated the use of dentifrice with strontium salt can provide subjective relief.54,55
Arginine and calcium carbonate are also used to address hypersensitivity. When arginine is combined with a bicarbonate buffer and calcium carbonate, it precipitates into exposed tubules, occluding them. The positive charge of the calcium carbonate-arginine complex facilitates precipitation on the negatively charged dentinal surface and tubule occlusion.56 The alkalinity of the arginine and calcium carbonate has also been found to increase uptake of calcium and phosphate ions into saliva, which are then deposited on dentin.56 The occlusion caused by arginine and calcium carbonate dentinal tubule plugs has been shown to be resistant to dietary acid exposure and pulpal pressures.57,58 Reductions in sensitivity noted with arginine and calcium carbonate occur immediately after application and persist for up to 28 days.32,56,57 A recent systematic review also reported this combination provided greater sensitivity relief than strontium acetate.59
Calcium sodium phosphosilicate exchanges sodium ions with hydrogen ions, allowing for the release of calcium and phosphate from the compound within the dentifrice. These minerals deposit within dentinal tubules, resulting in occlusion.60 Calcium sodium phosphosilicate has been shown to be effective in reducing sensitivity,61,62 however, there are equivocal data regarding the effectiveness of calcium sodium phosphosilicate compared to arginine and calcium carbonate. While the latter has been shown to result in deeper dentinal tubule plugs compared to the former,60 an in vivo study demonstrated a greater reduction in sensitivity with calcium sodium phosphosilicate.63 A major difference in the method of application of the dentifrices between these studies included the use of an electric toothbrush in the calcium sodium phosphosilicate study, which may have enhanced or altered uptake of the agents.60,62
Stannous fluoride and sodium fluoride in high concentrations are effective in managing sensitivity.64–67 These are usually prescription-only fluoride dentifrice or gels that can deliver up to 5000 to 12,500 ppm fluoride. The relief provided by these agents may be due to an increase in tubule occlusion and a reduction in acid decalcifications.68,69 Fluoride has an additional advantage of remineralization of dental tissues and reduction in susceptibility to radicular caries. If patients also have common risk factors for caries, this may be of increased importance for individuals with gingival recession and resultant root sensitivity following periodontal therapy.70,71
PROFESSIONALLY APPLIED DESENSITIZERS
In-office dentin desensitizers are often used in combination with patient-delivered desensitizers (e.g., dentifrice) and may contain fluoride, arginine and calcium carbonate, bonding agents, glutaraldehyde, oxalate or casein phosphopeptide/amorphous calcium phosphate. These agents may be used as monotherapy or in combination with other treatments.
Fluoride varnish is a desensitizer and commonly applied onto exposed, symptomatic root surfaces. The varnish solution interacts with saliva, which increases its stability on the tooth surface and allows increased fluoride uptake. Fluoride varnish has been shown to reduce sensitivity and provide longer-lasting relief than potassium nitrate.72 Varnish containing 5% sodium fluoride has been shown to be effective for up to 24 weeks after application.73
Arginine and calcium carbonate are available in a paste that can be applied to exposed root surfaces to treat dentinal hypersensitivity. A recent systematic review concluded that studies evaluating this technology demonstrate its efficacy in reducing sensitivity. However, the current body of evidence does not allow an assessment of the comparative effectiveness of arginine and calcium carbonate compared to other professionally administered topical desensitizers.74
Bonding agents, such as methacrylate resins and self-etch bonding systems containing both acidic ingredients to condition the dentin and monomers that cover dentinal tubules, have been used as desensitizing agents.75 In general, they provide fairly long-lasting relief,76 but sensitivity is likely to return if the resin breaks away and reexposes the tubules. This technique may be best used in recalcitrant cases of localized sensitivity, rather than generalized dentinal pain.77
Glutaraldehyde is used alone or with hydroxyethyl methacrylate (HEMA) as a desensitizing varnish. The presence of HEMA acts as a wetting agent to allow better adhesion of the glutaraldehyde to the tooth surface,78 where it reacts with serum albumin in saliva and crevicular fluid, resulting in precipitation of the serum albumin. This reaction causes polymerization of the HEMA and blockage of dentinal tubules.42,79,80 Modest-to-moderate reductions in sensitivity has been reported with glutaraldehyde alone, but the addition of HEMA increases efficacy and may prolong the duration of action up to nine months.81,82
Oxalate is a professionally applied desensitizer that combines with calcium ions in saliva to form insoluble calcium oxalate crystals that precipitate and block dentinal tubules.76,83,84 Acid etching prior to application may allow for deeper penetration into the tubules.85 Oxalate has been shown to reduce sensitivity76,86 and remains stable in an acidic environment, which could make it more durable and/or long-lasting than some other topical agents.76
Casein derivatives, such as casein phosphopeptide/amorphous calcium phosphate and other casein-derived peptides, bind to exposed dentin surfaces and allow for deposition of minerals from oral fluids, which decreases tubule openings. These substances provide bioavailable calcium and phosphate that can be released during intraoral acidic challenges and may encourage tooth remineralization.85 Studies on the efficacy of casein derivatives on hypersensitivity are equivocal, and additional standardized research may allow for assessment of their relative potency in reducing sensitivity.74,87
LASER THERAPY
Lasers, such as neodymium-doped yttrium aluminium garnet, erbium yttrium aluminium garnet, diode and CO2, have been studied for use in managing sensitivity.88 While these lasers (at the wavelengths and protocols tested) did not permanently alter dentin surfaces through melting the dentin to occlude dentinal tubules, it is believed they may reduce sensitivity through increased coagulation of proteins, which precipitate to block the tubules.89 Other laser technologies, such as helium-neon and gallium aluminium arsenate, have also been investigated to determine their effect on nerve activity.90–92 Research has demonstrated a reduction in root sensitivity after periodontal surgery in cases in which low-level irradiation was performed.93 A systematic review concluded that lasers are effective, but the current evidence is considered weak.87 Additionally, researchers have cautioned the risk for a placebo effect may be high when evaluating laser therapy.88,94 This modality also appears to increase the effectiveness and duration of action of traditional methods of tubule occlusion using topical therapies.94 While lasers may hold promise for managing posttreatment root sensitivity, further studies are needed to identify protocols for their safe and efficacious use.
CERVICAL RESTORATIONS
Cervical restorations may be used to treat dentinal hypersensitivity by covering tubules with restorative resins. This approach has demonstrated greater efficacy than patient-applied dentifrices — and has a more immediate effect, as well.95 Although research has demonstrated similar relief with cervical restorations or gingival root coverage grafts, Leybovich et al96 report that patients prefer gingival grafting for esthetic reasons. Cervical restorations are an invasive approach that requires irreversible preparation to achieve retention. In addition, marginal overhangs and leakage can lead to recurrent caries and/or gingival irritation from plaque. The use of fluoride-releasing glass ionomer materials may be considered for patients at high caries risk.97 Generally, cervical restorations should be considered only after failure of less-invasive therapy.
PERIODONTAL ROOT COVERAGE
Soft tissue grafting procedures can reduce sensitivity at sites with gingival recession and offer the advantage of increasing periodontal root coverage — and potentially increasing the width of keratinized gingiva.96,98,99 It is important to note that soft tissue grafting is most effective in areas of buccal gingival recession, and the predictability of root coverage is dependent upon the level of interproximal bone.100 A thorough evaluation is needed to determine if the clinical presentation after periodontal therapy is a site amenable to achieving root coverage. In most patients undergoing periodontal therapy, attachment loss would result in an environment in which complete root coverage may not be anticipated. In these cases, root coverage procedures can be combined with other restorative and/or desensitizing techniques or alternative care. As root coverage is an effective method for reducing sensitivity, it should be prioritized at sites where it could prove beneficial. Careful assessment and diagnosis of areas with root sensitivity are crucial when selecting patients for surgical therapy who are likely to achieve positive outcomes based upon their attachment levels and gingival recession morphology.
ROOT CANAL THERAPY
Root canal therapy is generally used to treat irreversible pulpitis and pulpal necrosis, and the procedure eliminates all sensory feeling associated with the tooth.95 While this would certainly eliminate dentinal hypersensitivity, except in extreme cases, this condition is not an indication for endodontic treatment. Given its invasive nature, root canal therapy should only be considered when no other option can eliminate sensitivity.38
CONCLUSION
Dentinal hypersensitivity is a common condition and is prevalent in patients who have received periodontal therapy.5–10 While many cases of root sensitivity resolve spontaneously within a relatively short time period,8 it is important to address patient discomfort following periodontal procedures — and in cases that persist well after completion of therapy. Clinicians should take a systematic approach to treatment and select therapies based on the diagnosis and etiologies identified during patient assessment. It is beneficial to consider less invasive, first-line therapies before attempting more invasive procedures to treat dentinal hypersensitivity.
REFERENCES
- Chabanski MB, Gilliam DG. Aetiology, prevalence, and clinical features of cervical dentine sensitivity. J Oral Rehabili. 1997;24:15–19.
- Holland GR, Nähri MN, Addy M, Gangarosa L, Orchardson R. Guidelines for the design and conduct of clinical trials on dentine hypersensitivity. J Clin Periodontaol.1997;24:808–813.
- Araha AC, Pimenta LA, Marchi GM. Clinical evaluation of desensitizing treatments for cervical dentin hypersensitivity. Braz Oral Res. 2009;23:333–339.
- Addy M, Pearce N. Aetiological, predisposing and environmental factors in dentine hypersensitivity. Arch Oral Biol. 1994;39:33S–38S.
- Badersten A, Nilvèus R, Egelberg J. Effect of non-surgical periodotnal therapy (II). Severely advanced periodontitis. J Clin Periodontol. 1984;11:63–76.
- Bissada NF. Symptomatology and clinical features of hypersensitive teeth. Arch Oral Biol. 1994;39 (Suppl):31S–32S.
- Haugen E, Johansen JR. Tooth hypersensitivity after periodontal treatment. A case report including SEM studies. J Clin Periodontol. 1988;15:399–401.
- von Troil B, Needleman I, Sanz M. A systematic review of the prevalence of root sensitivity following periodontal therapy. J Clin Periodontol. 2002;29 (Suppl 3):173–177.
- Tammaro S, Wennström JL, Bergenholtz G. Root-dentin sensitivity following non-surgical periodontal treatment. J Clin Periodontol. 2000;27:690–697.
- Van Volkinburg JW, Green E, Armitage GC. The nature of root surfaces after curette, cavitron, and alpha-sonic instrumentation. J Periodontal Res. 1976; 11:374–381.
- West NX, Lussi A, Seong J, Hellwig E. Dentin hypersensitivity: pan mechanisms and aetiology of exposed cervical dentin. Clin Oral Investig. 2013;17: S9–S19.
- Brännström M. A hydrodynamic mechanism in the transmission of pain-producing stimuli through the dentine. In: Anderson DJ, ed. Sensory Mechanisms in Dentine. Oxford: Pergamon Press;1963:73–79.
- Irvine JH. Root surface sensitivity: a review of aetiology and management. J N Z Soc Periodont. 1988;66:15–17.
- Dababneh RH, Khouri AT, Addy M. Dentine hypersensitivity — an enigma? A review of terminology, mechanisms, aetiology, and management. Br Dent J. 1999;187:606–611.
- Addy M. Dentine hypersensitivity: New perspectives on an old problem. Int Dent J. 2002; 52 (Suppl):367–375.
- Absi EG, Addy M, Adams D. Dentine hypersensitivity. A study of the patency of dentinal tubules in sensitive and non-sensitive cervical dentine. J Clin Periodontol. 1987;14:280–284.
- Yoshiyama M, Masada J, Uchida A, Ishida H. Scanning electron microscopic characterization of sensitive vs. insensitive human radicular dentine. J Dent Res. 1989;68:1498–1502.
- Rees JS, Addy M. A cross-sectional study of dentine hypersensitivity. J Clin Periodontol. 2002;29: 997–1003.
- Gilliam DG, Seo H, Bulman JS, Newman HN. Perceptions of dentine hypersensitivity in a general practice population. J Oral Rehabil. 1999;26:710–714.
- Fischer C, Fischer RG, Wennberg A. Prevalence and distribution of cervical dentine hypersensitivity in a population in Rio de Janeiro, Brazil. J Dent. 1992; 20:272–276.
- Taani SD, Awartani F. Clincial evaluation of cervical dentin sensitivity (CDS) in patients attending general dental clinical (GDC) and periodontal specialty clinics (PSC). J Clin Periodontol. 2002;29: 118–122.
- Pashley DH. Dentine permeability: Theory and practice. In: Spangberg LSW, ed. Experimental Endodontics. Boca Raton, FL: CRC Press; 1990:19–49.
- Konturri-Närhi V, Närhi M. Factors related to the occurrence of pain symptoms 0.5–5 years after periodontal surgery. J Dent Res. 1998;77:672.
- Jensen AL. Hypersensitivity controlled by iontophoresis: double-blind clinical investigation. J Am Dent Assoc. 1964;68:216–225.
- Graf H, Galasse R. Morbidity, prevalence and intraoral distribution of hypersensitive teeth. J Dent Res. 1977;53 (Spec Issue A):162.
- Gilliam DG, Seo HS, Newman HN, Bulman JS. Comparison of dentine hypersensitivity in selected occidental and oriental populations. J Oral Rehab. 2001;28:20–25.
- Blaggana A, Vohra P, Nagpal A. Diagnosis and treatment of dentinal hypersensitivity. J Innovative Dent. 2011;1:1–4.
- Chu CH, Lui KS, Lau KP, Kwok CM, Huang T. Effects of 8% arginine desensitizing paste on teeth with hypersensitivity. J Dent Res. 2010;89(Spec Issue A).
- Wallace JA, Bissada NF. Pulpal and root sensitivity rated to periodontal therapy. Oral Surg Oral Med Oral Pathol. 1990;69:743–747.
- Noyes FB, Schour I, Noyes HJ. Textbook of Dental Histology and Embryology. 5th ed. Philadelphia: Lea & Febiger; 1938.
- Kim SY, Kim EJ, Kim DS, Lee IB. The evaluation of dentinal tubule occlusion by desensitizing agents: A real-time measurement of dentinal tubule flow rate and scanning electron microscopy. Oper Dent. 2013;38:419–428.
- Panagakos F, Schiff T, Guignon A. Dentin hypersensitivity: effective treatment with an in-office desensitizing paste containing 8% arginine and calcium carbonate. Am J Dent. 2009;22:3A–7A.
- Kawasaki A, Ishikawa K, Suge T, et al. Effects of plaque control on the patency and occlusion of dentine tubules in situ. J Oral Rehabil. 2001;28:439–449.
- Addy M. Mostafa P, Newcombe G. Dentine hypersensitivity: the distribution of recession, sensitivity, and plaque. J Dent. 1987;15:242–248.
- Bender IB. Pain conference summary. J Endod. 1986;12:509–517.
- Manochehr-Pour M, Bhat M, Bissada N. Clinical evaluation of two potassium nitrate toothpastes for the treatment of dentinal hypersensitivity. Periodontal Case Rep. 1984;6:25–30.
- Haneet RK, Vandana LK. Prevalence of dentinal hypersensitivity and study of associated factors: a cross-sectional study based on the general dental population of Davangere, Karnataka, India. Int Dent J. 2016;66:49–57.
- Clark D, Levin L. Non-surgical management of tooth hypersensitivity. Int Dent J. 2016;66:249–256.
- Matis BA, Cochran MA, Eckert GJ, Mathis JI. In vivo study of two carbamide peroxide gels with different desensitizing agents. Oper Dent. 2007;32: 549–555.
- Leonard RH Jr, Smith LR, Garland GE, Caplan DJ. Desensitizing agent efficacy during whitening in an at-risk population. J Esthet Restor Dent. 2004;16: 49–55.
- Chu CH, Lam A, Lo EC. Dentin hypersensitivity and its management. Gen Dent. 2011;59:115–122.
- Porto IC, Andrade AK, Montes MA. Diagnosis and treatment of dentinal hypersensitivity. J Oral Sci. 2009;51:323–332.
- Markowitz K, Kim S. Hypersensitive teeth. Experimental studies of dentinal desensitizing agents. Dent Clin North Am. 1990;34:491–501.
- Ayad F, Berta R, De Vizio W, McCool J, Petrone ME, Volpe AR. Comparative study of two dentifrices containing 5% potassium nitrate on dentinal sensitivity: a twelve-week clinical study. J Clin Dent. 1994;5:97–101.
- Schiff T, Dotson M, Cohen S, De Vizio W, McCool J, Volpe A. Efficacy of a dentifrice containing potassium nitrate, soluble pyrophosphate, PVM/MA copolymer and sodium fluoride on dentinal hypersensitivity: a twelve-week clinical study. J Clin Dent. 1994;5:87–92.
- Frechoso SC, Menendez M, Guisasola C, Arregui I, Tejerina JM, Sicilia A. Evaluation of the efficacy of two potassium nitrate bioadhesive gels (5% and 10%) in the treatment of dentine hypersensitivity. A randomized controlled trial. J Clin Periodontol. 2003; 30:315–320.
- Gillam DG, Bulman JS, Jackson RJ, Newman HN. Comparison of 2 desensitizing dentifrices with a commercially available fluoride dentifrice in alleviating cervical dentine sensitivity. J Periodontol. 1996;67:737–742.
- West NX, Addy M, Jackson RJ, Ridge DB. Dentine hypersensitivity and the placebo response. A comparison of the effect of strontium acetate, potassium nitrate, and fluoride toothpastes. J Clin Periodontol. 1997;24:209–215.
- Poulsen S, Errboe M, Hovgaard O, Worthington HW. Potassium nitrate toothpaste for dentine hypersensitivity. Cochrane Database Syst Rev. 2001; 2:CD001476.
- Tarbet WJ, Buckner A, Stark MM, Fratarcangelo PA, Augsburger R. The pulpal effects of of brushing with a 5 percent potassium nitrate paste used for desensitization. Oral Surg Oral Med Oral Pathol. 1981;51:600–602.
- Olley RC, Moazzez R, Bartlett D. Effects of dentifrices on subsurface dentin tubule occlusion: an in situ study. Int J Prosthodont. 2015;28:181–187.
- Kun L. Biophysical study of dental tissues under the effect of a local strontium application. Schweiz Monatsschr Zahnheilkd. 1976;86:661–676.
- Mishima H, Sakae T, Kozawa Y. Scanning electron microscopy and energy dispersive spectroscopy analysis of calciotraumatic lines in rat labial dentin after acute exposure of strontium chloride. Scanning Microsc. 1995;9:797–803.
- Pearce NX, Addy M, Newcombe RG. Dentine hypersensitivity: a clinical trial to compare 2 strontium desensitizing toothpastes with a conventional fluoride toothpaste. J Periodontol. 1994;65:113–119.
- Markowitz K, Pashley DH. Discovering new treatments for sensitive teeth: the long path from biology to therapy. J Oral Rehabil. 2008;35:300–315.
- Kleinberg I. Sensistat. A new saliva-based composition for simple and effective treatment of dentinal sensitivity pain. Dent Today. 2002;21:42–47.
- Cummins D. Dentin hypersensitivity: from diagnosis to breakthrough therapy for everyday sensitivity relief. J Clin Dent. 2009;20:1–9.
- Petrou I, Heu R, Stanick M, et al. A breakthrough therapy for dentin hypersensitivity: how dental products containing 8% arginine and calcium carbonate work to deliver effective relief of sensitive teeth. J Clin Dent. 2009;20:23–31.
- Magno MB, Nascimento GC, Da Penha NK, et al. Difference in effectiveness between strontium acetate and arginine-based toothpastes to relieve dentin hypersensitivity. A systematic review. Am J Dent. 2015;28:40–44.
- Chen CL, Parolia A, Pau A, Celerino de Moraes Porto IC. Comparative evaluation of the effectiveness of desensitizing agents in dentinal tubule occlusion using scanning electron microsopy. Aus Dent J. 2015;60:65–72.
- Pradeep AR, Sharma A. Comparison of clinical efficacy of a dentifrice containing calcium sodium phosphosilicate to a dentifrice containing potassium nitrate and to a placebo on dentinal hypersensitivity: a randomized clinical trial. J Periodontol. 2010;81:1167–1173.
- Rajesh KS, Hedge S, Arun Kumar MS, Shetty DG. Evaluation of the efficacy of a 5% calcium sodium phosphosilicate (Novamin®) containing dentifrice for the relief of dentinal hypersensitivity: a clinical study. Indian J Dent Res. 2012;23:363–367.
- West NX, Macdonald EL, Jones SB, Clayton NC, Hughes N, Jeffery P. Randomized in situ clinical study comparing the ability of two new desensitizing toothpaste technologies to occlude patent dentin tubules. J Clin Dent. 2011;22:82–89.
- Minkov B, Marmari I, Gedalia I, Garfunkel A. The effectiveness of sodium fluoride treatment with and without iontophoresis on the reduction of hypersensitive dentin. J Periodontol. 1975;46:246–249.
- Gedalia I, Bayer L, Kalter N, Richter M, Stabhjolz A. The effect of sodium fluoride and strontium application on dentine: in vivo and in vitro studies. J Periodontol. 1978;49:269–272.
- Kerns DG, Scheidt MJ, Pashley DH, Horner JA, Strong SL, Van Dyke TE. Dentinal tubule occlusion and root hypersensitivity. J Periodontol. 1991;62:421–428.
- Miller JT, Shannon IL, Kilgore WG, Bookman JE. Use of water-free stannous fluoride containing gel in the control of dentinal hypersensitivity. J Periodontol. 1969;40:490–491.
- Tal M, Oron M, Gedalia I, Ehrlich J. X-ray diffraction and scanning electron microscope investigations of fluoride-treated dentine in man. Arch Oral Biol. 1976;21:285–290.
- Fuhseth R. A study of experimentally exposed and fluoride treated dental cementum in pigs. Acta Odont Scan. 1970;28:833–850.
- O’Mullane DM, Baez RJ, Jones, S, et al. Fluoride and oral health. Community Dent Health. 2016;33: 69–99.
- Marinho VC, Higgins JP, Sheiham A, Logan S. Combinations of topical fluoride (toothpastes, mouthrinses, gels, vernishes) versus single topical fluoride for preventing dental caries in children and adolescents. Cochrane Database Syst Rev. 2004;1:CD002781.
- Pandit N, Gupta R, Bansal A. Comparative evaluation of two commercially available desensitizing agents for the treatment of dentinal hypersensitivity. Indian J Dent Res. 2012;23:778–783.
- Ritter AV, de L Dias W, Miguez P, Caplan DJ, Swift EJ Jr. Treating cervical dentin hypersensitivity with fluoride varnish: a randomized clinical study. J Am Dent Assoc. 2006;137:1013–1020.
- West NX, Seong J, Davies M. Management of dentine hypersensitivity: efficacy of professionally and self-administered agents. J Clin Periodontol. 2015;42 (Suppl 16):S256–S302.
- Brannstrom M, Johnson G. Effects of various conditioners and cleansing agents on prepared dentin surfaces: a scanning electron microscopic investigation. J Prosthet Dent. 1974;31:422–430.
- Pashley DH, Galloway SE. The effects of oxalate treatment on the smear layer of ground surfaces of human dentine. Arch Oral Biol. 1985;30:731–737.
- Addy M, Dowell P. Dentine hypersensitivity — a review. Clinical and in vitro evaluation of treatment agents. J Clin Periodontal. 1983;10:351–363.
- Stewardson DA, Crisp RJ, McHugh S, Lendenmann U, Burke FJ. The effectiveness of Systemp.desensitizer in the treatment of dentine hypersensitivity. Prim Dent Care. 2004;11:71–76.
- Qin C, XU J, Zhang Y. Spectroscopic investigation of the function of aqueous 2-hydroxythymethacrylate/glutaraldehyde solotion as a dentin desentizer. Eur J Oral Sci. 2006;114:354–359.
- Schüpbach P, Lutz F, Finger WJ. Closing of dentinal tubules by Gluma desensitizer. Eur J Oral Sci. 1997;105:414–421.
- Kakaboura A, Rahiotis C, Thomaidis S, Doukoudakis S. Clinical effectiveness of two agents on the treatment of tooth cervical hypersensitivity. Am J Dent. 2005;18:291–295.
- Patil SA, Naik BD, Suma R. Evaluation of three different agents for in-office treatment of dentinal hypersensitivity: A controlled clinical study. Indian J Dent Res. 2015;26:38–42.
- Pashley DH. Dentin permeability, dentin sensitivity, and treatment through tubule occlusion. J Endod. 1986;12:465–474.
- Moncada G, Fernandez E, Deyer E, et al. In-vivo study of calcium oxalate as a root surface desensitizer. Presented at IADR/AADR/CADR 83rd General Session. Baltimore, MD; March 9–12, 2005.
- Trushkowsky RD, Oquendo A. Treatment of dentin hypersensitivity. Dent Clin North Am. 2011;55:599–608.
- Pashley DH, Livingston MJ, Reeder OW, Horner J. Effects of the degree of tubule occlusion on the permeability of human dentine in vitro. Arch Oral Biol. 1978;23:1127–1133.
- Kowalczyk A, Botulinski B, Jaworska M, Kierklo A, Pawinska M, Dabrowska E. Evaluation of the product based on Recaldent technology in the treatment of dentin hypersensitivity. Adv Med Sci. 2006;51 (Suppl 1):40–42.
- Sgolastra F, Petrucci A, Gatto R, Monaco A. Effectiveness of laser in dentinal hypersensitivity treatment: a systematic review. J Endod. 2011;37:297–303.
- Goodis, HE, White JM, Marshall SJ, et al. Measurement of fluid flow through laser-treated dentine. Arch Oral Biol. 1994;9:128S.
- Rochkind S, Nissan M, Razon N, Schwartz M, Bartal A. Electrophysiological effect of HeNe laser on normal and injured sciatic nerve in the rat. Acta Neurochir (Wien). 1986; 83:125–130.
- Rochkind S, Nissan M, Bar-Nea L, Razon N, Schwartz M, Bartal A. Response of peripheral nerve to He-Ne laser: Experimental studies. Lasers Surg Med. 1987;7:441–443.
- Wakabayashi H, Hamba M, Matsumoto K, Nakayama T. Electrophysiological study of irradiation of semiconductor laser on the activity of the trigeminal subnucleues caudal neurons. J Jpn Soc Laser Dent. 1992;3:65–74.
- Shreya D, Sanjay J, Rashmi H. Effect of low-level laser therapy in reducing dentinal hypersensitivity and pain following periodontal flap surgery. Photomed Laser Surg. 2014;32:700–706.
- Biagi R, Cossellu G, Sarcina M, Pizzamiglio IT, Farronato G. Laser-assisted treatment of dentinal hypersensitivity: a literature review. Ann Stomatol (Roma). 2015;6:75–80.
- Veitz-Keenan A, Barna JA, Strober B, et al. Treatment for hypersensative noncarious cervical lesions: A Pratitioners Engaged in Applied Research and Learning (PEARL) Network randomized clinical effectiveness study. J Am Dent Assoc. 2013;144:495–506.
- Leybovich M, Bissada NF, Teich S, Demko CA, Ricchetti PA. Treatments of noncarious cervical lesions by a subepthelial connective tissue graft versus a composite resin restoration. Int J Periodontics Restorative Dent. 2014;649–654.
- Gordan VV, Blaser PK, Watson RE, et al. A clinical evaluation of a giomer restorative system containing surface prereacted glass ionomer filler: results from a 13-year recall examination. J Am Dent Assoc. 2014; 145:1036–1043.
- Douglas de Oliveira DW, Marques DP, Anguiar-Cantuária IC, Flecha OD, Goncalves PF. Effect of surgical defect coverage on cervical dentin hypersensitivity and quality of life. J Periodontol. 2013;84:768–775.
- Chambrone L, Sukekava F, Araújo MG, Pustiglioni FE, Chambrone LA, Lima LA. Root coverage procedures for the treatment of localized recession-type defects: a Cochrane systematic review. J Periodontol. 2010;81:452–478.
- Miller PD Jr. A classification of marginal tissue recession. Int J Periodontics Restorative Dent. 1985; 5:8–13.
FEATURED IMAGE BY RYANKING999/ISTOCK/THINKSTOCK
From Decisions in Dentistry. May 2017;3(5):37—42.




