
Examining for Periodontal Disease
These clinical strategies will help reveal pathology in its early or advanced stages.
Inflammatory periodontal disease ranks among the most common chronic infections in humans.1 These infections are responsible for a large percentage of tooth loss in adults.2 In addition, the ubiquity and recurrence of periodontal disease, and the damage it causes, require frequent examinations to prevent — and, when indicated, treat — these conditions. Examining and recording data on the state of the periodontium should therefore be an integral part of the routine information gathered on every dentate patient. The emphasis of this article is on what constitutes an appropriate periodontal examination, the timing and extent of data collection, and how clinicians can determine end points in therapy based on this information.
The severity and course of periodontal disease can be profoundly affected by the patient’s systemic health. There is also accumulating evidence that chronic infections of all types, including periodontal conditions, can have negative systemic health effects.3 Therefore, gathering information on the patient’s overall health is an integral part of the clinical data. Questions about smoking and diabetes are especially important because both are significant risk factors for periodontal disease.4,5 Medications currently taken, along with drug allergies, should also be recorded. Additional medical history data should include demographics and familial medical/dental history, as well as baseline vitals, height and weight. Any unanswered questions related to the patient’s systemic health should be addressed in writing to the individual’s physician.
An overview of the head and neck, including muscles of mastication, the temporomandibular joints and an examination for any inappropriate extraoral findings, should be performed. This is followed by an intraoral examination designed to identify and record any abnormalities.
BEHAVIORAL CHANGES
If periodontal disease is present, the goal of therapy is restoring health and function to these tissues. Because these conditions are chronic and the oral biofilm responsible for their initiation constantly returns, patient motivation to remove biofilm on a daily basis is vitally important. This can require significant behavioral changes on the patient’s part. These changes are difficult to initiate and even harder to sustain. In addition, it should be understood that the patient’s desires and goals might not always be aligned with those of the dental provider. Thus, it is important for clinicians to understand the patient’s concerns and goals.
Treatment therefore begins with listening to the patient, as he or she will usually provide information that is key to motivating behavioral change needed to control periodontal disease. This can also help the provider understand the degree of knowledge that the patient has concerning the disease process. Patients are more likely to undergo positive behavioral changes that are designed to help them solve current problems and reach their goals. In most cases, providing detailed information concerning the nature of the disease, possible therapeutic alternatives, and how they benefit the patient, will help instigate and sustain positive behavioral changes.
BASELINE MEASUREMENTS
In periodontal therapy, it is important to collect data to establish a baseline. This material will help clinicians decide what initial treatment is appropriate and also can be used to judge the patient’s response to therapy. The following is a list of examination parameters that will provide adequate evidence about the patient’s periodontal status to make an initial diagnosis and develop a treatment plan.
A full mouth series of periapical radiographs and posterior vertical bitewings are preferred to a panoramic radiograph with horizontal bitewings, as periapical radiographs exposed at right angles to the teeth provide more accurate and detailed information on the position of the interproximal bone. As opposed to horizontal bitewings, the use of vertical posterior bitewings is suggested because these films provide more accurate information concerning interproximal bone loss and the relationship of the crest of the alveolar bone to the cementoenamel junction (CEJ). When evaluating these films, it should be remembered that the interproximal crest of bone is normally 2 mm apical to the CEJ. Any abnormalities or anatomical anomalies should be recorded.6
A count of missing teeth, as well as their replacements, should be recorded; this includes any supernumerary or retained deciduous teeth. A search for improper tooth-to-tooth relations, tilted, rotated or supra-erupted teeth is necessary, as these malpositions can lead to biofilm retention and subsequent periodontal problems. These abnormalities should be recorded and eliminated whenever possible.
Open or irregular interproximal contacts, marginal ridge discrepancies, plunger cusps and similar conditions can lead to food impaction and subsequent bone loss, and should be addressed. The genesis of these problems is often occlusal. Accordingly, appropriate evaluation and subsequent elimination of these problems can help reduce future bone loss.
Occlusal abnormalities, including wear facets (which can also indicate occlusal trauma), should be evaluated. A general examination of occlusion — including the distance between centric relation (CR) and centric occlusion, as well as which teeth occlude in working excursions — is a key step. In addition, identifying Angle’s malocclusion is helpful in diagnosis and treatment planning.
Compared to teeth without such contacts, Harrel and Nunn7 have shown that premature contacts in CR and nonworking occlusal contacts can increase periodontal attachment loss over time. These findings may suggest the need for occlusal adjustment and/or fabrication of habit devices (such as night guards) for patients being treated for periodontal disease.
BIDIGITAL TOOTH MOBILITY
Bidigital tooth mobility is measured using the nonworking ends of a dental mirror and periodontal probe. A common quantitation of mobility involves a modification of the Miller scale, as recommended by Fleszar et al8 (Figure 1).
- Class 0: physiologic mobility; firm tooth
- Class I: Slightly increased mobility
- Class II: Definite-to-considerable increase in mobility, but no impairment of function
- Class III: Extreme mobility; a loose tooth that would be uncomfortable in function
It should be noted the use of this scale can result in a great deal of variation between individual examiners, as well as between offices; with practice, however, a degree of reproducibility can be found. And while considerable controversy exists, most practitioners would suggest the longevity of a tooth that is stable is greater than one that is mobile.
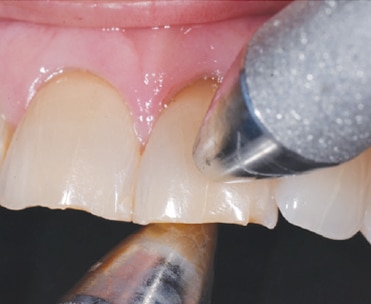
FREMITUS
Fremitus is defined as tooth movement during function, and there are two ways to measure this parameter. The first is visual. The patient is asked to tap his or her teeth together, and grind from side to side as pressure from the clinician’s hand keeps the teeth in contact. Any tooth movement seen is fremitus. The second method is tactility. The clinician’s fingers are placed on the facial aspect of the teeth in the maxillary arch, and the patient is instructed to hold the teeth tightly together and guided in excursive movements under pressure. Any tooth movement is fremitus. The authors have found that many hypersensitive teeth have fremitus, and that sensitivity is ameliorated following elimination of this parameter using selective occlusal adjustment.
SULCUS/POCKET PROBING DEPTHS
Recording pocket probing depths around each tooth is a requisite during a periodontal examination. Six readings are taken around each tooth. A periodontal probe of 1-mm diameter with Williams or North Carolina markings is suggested, as is a probing force of 15 Newtons (15.30 grams). With practice, reproducible measurements can be obtained. Pocket probing depths of 1 to 3 mm are considered normal. This assumes the margins of the gingival tissues are at their normal level; that is, slightly coronal to the CEJ (Figure 2).
Any bleeding seen within 10 to 15 seconds after removing the probe from the pocket should be recorded. Studies have shown that absence of bleeding on probing indicates that a state of health will usually continue for several months after the examination.9 Although the significance of bleeding on probing in terms of disease progression is still not completely understood, as previously stated, recent data suggest that chronic inflammation of all types, including periodontal disease, can negatively affect systemic health.3 By extension, this indicates that collection of probing data is appropriate. This is because bleeding on probing is generally considered a sign of inflammation. Reduction or elimination of bleeding should be a key goal of therapy.
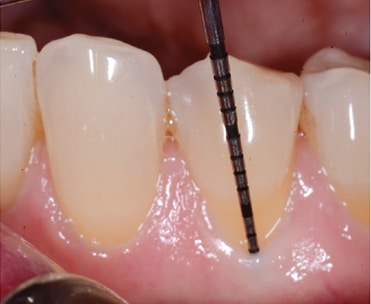
GINGIVAL RECESSION
During the periodontal examination, buccal, lingual and interproximal tissue heights should recorded at a fixed point (usually the CEJ or the margin of a restoration). Recession is recorded as positive when the gingival margin is located apical to the CEJ, and as negative when tissue is above the CEJ (Figure 2).
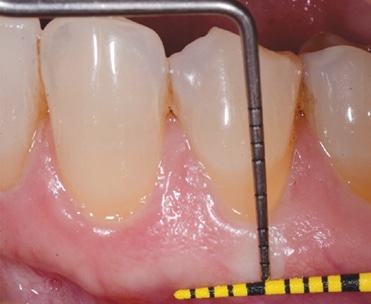
KERATINIZED AND ATTACHED GINGIVA
There is evidence that it is not absolutely necessary to have attached gingiva around teeth to maintain health. However, clinical experience has shown there is less recession where there is 1 to 2 mm of attached gingiva
Normal gingival tissue is firm and stippled. The coronoapical dimension width of the keratinized gingiva is measured on the midfacial and midlingual of the free gingival margin to the mucogingival junction. The amount of attached gingiva is determined by subtracting the pocket probing depth from the amount of keratinized gingiva. The facial and lingual dimension of this tissue should be recorded for each tooth (Figure 3).
CLINICAL ATTACHMENT LEVEL
Clinical attachment level (CAL) is calculated by adding pocket probing depth to the amount of recession in each area around the tooth. These data points provide a more accurate representation of the amount of attachment loss around each surface of each tooth than probing depth alone. It is possible to have probing depths within normal range (1 to 3 mm), for example, and yet have CAL recordings of 10 to 12 mm, indicating very little attachment remaining around that surface of the tooth.
INITIAL DIAGNOSIS AND TREATMENT
At this point, an initial diagnosis is made. The options are health, gingivitis or periodontitis. Health is defined as no history of clinical attachment loss and no signs of inflammation. Gingivitis can be defined as probing depths of 1 to 3 mm with no history of attachment loss, but associated with clinical signs of inflammation — including color changes toward red and bleeding on probing. Periodontitis is defined as probing depths of 4 mm or greater, accompanied by clinical signs of inflammation in areas without gingival hypertrophy. The process is defined histologically by a loss of periodontal attachment apparatus (cementum, periodontal ligament and alveolar bone), resulting in an apical shift of the junctional epithelium and connective tissue attachment.10 While most patients with periodontitis have the chronic form, an aggressive form of this disease — characterized by rapid loss of attachment — can be seen in some individuals. This often presents in young patients and should be referred to a specialist. Notably, specific classifications of periodontal disease have been updated recently by the American Academy of Periodontology task force regarding CAL, localized versus generalized periodontitis, and chronic versus aggressive periodontitis.11
Treatment of these chronic infections includes appropriate instruction in personal hygiene and clinical removal of local factors (including supra- and subgingival calculus and biofilm). Any appropriate initial occlusal therapy is performed at this stage. Following treatment, a reevaluation of the patient’s periodontal status is necessary. Residual signs of active chronic periodontitis often require surgical intervention. In fact, aggressive periodontitis often responds best to surgical therapy.
END POINTS AND MAINTENANCE
Ideal end points of periodontal therapy include probing depths of 3 mm or less, no clinical signs of inflammation (e.g., no bleeding on probing), the margin of the soft tissues at or slightly occlusal to the CEJ, and functional tooth mobility. Realistic endpoints include minimal signs of inflammation as evidenced by reduced bleeding on probing (10% of sites or fewer), and probing depths and teeth that are stable over time. Failure to reach these endpoints should warrant reevaluation and further treatment until the periodontium is stable. If deeper probing depths remain or inflammation continues, surgery to regenerate periodontal attachment may be indicated.
Once a patient’s end points are stable, a final diagnosis is made and this will determine maintenance intervals. Individuals with a diagnosis of health (i.e., no attachment loss or gingivitis) can normally be seen once a year for supportive periodontal therapy. Individuals with a final diagnosis of gingivitis usually respond well to supportive periodontal therapy twice per year, while patients with a final diagnosis of periodontitis typically require maintenance visits three to four times a year. More aggressive forms of this disease, of course, will require more frequent maintenance intervals.12
KEY TAKEAWAYS
- Examining and recording data on the state of the periodontium should be an integral part of the routine information gathered on every dentate patient.
- When recording health histories, questions about smoking and diabetes are especially important because these are significant risk factors for periodontal disease.
- Treatment begins with listening to the patient, as he or she will usually provide information that is key to motivating the behavioral change needed to control periodontal disease.
- Bleeding on probing is generally considered a sign of inflammation, and reduction or elimination of bleeding should be one goal of therapy.
- Ideal end points of periodontal therapy include probing depths of 3 mm or less, no clinical signs of inflammation, the margin of the soft tissues at or slightly occlusal to the cementoenamel junction, and functional tooth mobility.
REFERENCES
- U.S. Department of Health and Human Services. National Center for Health Statistics. Third National Health and Nutrition Examination Survey, NHANES III Examination Data File. Available at: wwwn.cdc.gov/nchs/data/nhanes3/1a/readme.txt. Accessed April 17, 2017.
- Akhter R, Hassan NM, Aida J, Zaman KU, Morita M. Risk indicators for tooth loss due to caries and periodontal disease in recipients of free dental treatment in an adult population in Bangladesh. Oral Health Prev Dent. 2008;6:199–207.
- Eke PI, Dye BA, Wei L, Thornton-Evans GO, Genco RJ. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91:914–920.
- Mealey BL, Oates TW, American Academy of Periodontology. Diabetes mellitus and periodontal diseases. J Periodontol. 2006;77:1289–1303.
- Rivera-Hidalgo F. Smoking and periodontal disease: a review of the literature. J Periodontol. 1986;57:617–624.
- Tugnait A, Clerehugh V, Hirschmann PN. The usefulness of radiographs in diagnosis and management of periodontal diseases: a review. J Dent. 2000;28:219–226.
- Harrel SK, Nunn ME. The association of occlusal contacts with the presence of increased periodontal probing depth. J Clin Periodontol. 2009;36:1035–1042.
- Fleszar TJ, Knowles JW, Morrison EC, Burgett FG, Nissle RR, Ramfjord SP. Tooth mobility and periodontal therapy. J Clin Periodontol. 1980;7:495–505.
- Lang NP, Adler R, Joss A, Nyman, S. Absence of bleeding on probing an indicator of periodontal stability. J Clin Periodontol. 1990;17:714–721.
- Page RC, Schroeder HE. Pathogenesis of inflammatory periodontal disease. A summary of current work. Lab Invest. 1976;34:235–249.
- American Academy of Periodontology. Task force report on the update to the 1999 classification of periodontal diseases and conditions. J Periodontal. 2015;86:835–838.
- Cohen RE, Research, Science and Therapy Committee, American Academy of Periodontology. Position paper: periodontal maintenance. J Periodontal. 2003;74:1395–1401.
The authors have no commercial conflicts of interest to disclose.
FEATURED IMAGE BY WAVEBREAKMEDIA/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. May 2017;3(5):11–14.


![The controversy around adding fluoride to drinking water was recently highlighted by an article in the Wall Street Journal, which addressed its possible negative neurological effects on children.[1] The article cited recent studies on what levels of fluoride are needed to protect teeth without risking possible cognitive harm to prenatal children and infants. Link in bio.
[1] Data on link.
---
#dentistry #dentist #dental #smile #dentista #teeth #cosmeticdentistry #dentistryworld #dentalphotography #odonto #tooth #dentistrylife #orthodontics #dentalcare #dentalhygienist #dentalimplants #oralhealth #veneers #dentalstudent #dentalassistant #dentalclinic #dentistlife #dentalhygiene #teethwhitening #oralsurgery #dds #endodontics #continuingeducation #education](http://decisionsindentistry.com/wp-content/plugins/instagram-feed/img/placeholder.png)