The Clinical Use of Lasers in Modern Dentistry
Discover how dental lasers are redefining soft and hard tissue care, periodontal therapy, surgery, implant recovery, and patient comfort.
Today, dental lasers are used for both soft and hard tissue procedures. When evaluating dental lasers, three concepts should be considered. The first is the target tissue chromophore(s) for the specific laser wavelength. A chromophore is the substance that absorbs laser energy allowing work to be done to the intended tissue. In soft tissue, the primary chromophores are hemoglobin, melanin, and water. In teeth and bone, the primary chromophores are water and hydroxyapatite.
The second concept is the depth of tissue penetration of laser energy, which depends on the laser’s wavelength and the proportion of light absorbed in the target tissue. Laser energy absorbed in water has a very shallow depth of tissue penetration.
The third concept is the type of photo effect (photoacoustic or photothermal) that occurs when light waves are converted to working energy. Knowledge of this effect will help determine appropriate thermal relaxation intervals and tissue cooling methods.
Visible and near infrared wavelengths are not absorbed in water, penetrate more deeply, and are primarily suited for soft tissue and photobiomodulation (PBM) procedures. Alternatively, near and midinfrared wavelengths are absorbed in water, penetrate less deeply, and are equally adept in soft and hard tissue procedures. Table 1 provides information on which lasers are indicated for different procedures.
Periodontal Diseases
One of the most widely used applications for dental lasers is in the treatment of periodontal diseases. This multistep process includes scaling and root planing to debride and disinfect sulcular epithelium (neodymium-doped yttrium aluminum garnet [Nd:YAG], diode), remove calculus and smear layer on root surfaces (erbium-doped yttrium aluminum garnet [Er:YAG]), and promote fibrin clot formation and coagulation (Nd:YAG, diode) collectively promoting epithelial reattachment. Nd:YAG and diode lasers are minimally absorbed by water, allowing deep tissue penetration and efficient removal and disinfection of diseased epithelium through absorption by hemoglobin and melanin. The shock waves created in water molecules from Er:YAG laser energy assist in calculus removal and elimination of the smear layer on root surfaces.
When using Nd:YAG, longer pulse durations of 650 microseconds stimulate fibrin clot formation. When diseased tissue lining the pocket is eliminated with debridement and laser, the fibrin clot functions as a scaffold for new tissue growth.1
Surgery and implant recovery
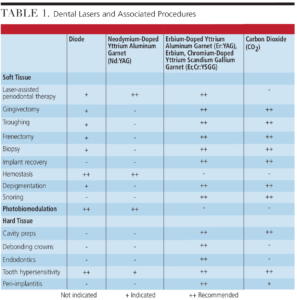
Creating a temporary space or mote around a crown or cavity preparation to isolate gingival margins is a routine procedure in the general dentist’s office. Lasers provide an attractive alternative to the placement of gingival retraction cord. To accomplish adequate troughing, the laser is used to surgically remove tissue while providing hemostasis. Erbium and carbon dioxide (CO2) lasers surgically remove tissue because of their absorption in water, but do not have the hemostatic effect that diode and Nd:YAG lasers provide. This is due to the absorption in hemoglobins and melanin and the photothermal tissue interaction that create coagulation. The result is a clean operating field with minimal trauma to the surrounding tissues and less postoperative discomfort. Figure 1 shows diode troughing and Figure 2 features CO2 troughing.
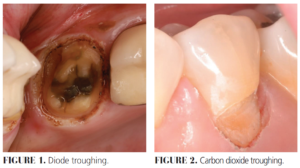
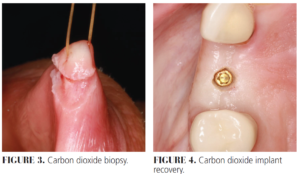
Lasers can be utilized for other surgical procedures such as frenectomies, gingivectomies, and biopsies. The precision of laser cuts promotes quick healing with less scarring and post-operative pain. Diode, Nd:YAG, and, to a lesser degree, CO2 lasers (Figure 3) coagulate as they cut, eliminating the need for sutures. When using erbium lasers for surgical procedures, additional methods for hemostasis should be considered. For biopsy procedures, the extent of collateral tissue damage must be managed and the type of laser used reported to the pathologist.
When uncovering a dental implant, a laser that will reflect off the implant’s metal surface while absorbed by soft tissue chromophores, such as water, hemoglobin, or melanin, is required. Erbium and CO2 lasers are reflected off metal and absorbed in water.2 In addition, the shallow depth of tissue penetration and minimal collateral damage make these lasers obvious choices for implant recovery (Figure 4).3 Photothermal tissue effects from diode and Nd:YAG lasers are dangerous to use around metal implants, as the heat generated can be fatal to osteocytes and cause irreversible thermal damage to the implant surface.4,5
Hemostasis
Oral health professionals encounter soft tissue bleeding routinely. The photothermal effects and absorption in hemoglobins and melanin make diode and Nd:YAG lasers highly effective for hemostasis and are frequently used in periodontal therapy. Erbium and CO2 lasers are designed for rapid tissue cutting due to their absorption in water. Only the CO2 laser’s photothermal effect coagulates blood vessels for adequate hemostasis and is often used in conjunction with operative procedures.
Depigmentation/De-epithelialization
Amalgam tattoos and melanin pigmentation can be removed with diode, Nd:YAG, erbium, and CO2 lasers. Diode and Nd:YAG laser energy is absorbed by dark pigments, resulting in tissue ablation. Conversely, erbium and CO2 light energy is absorbed by water in soft tissues, and the affected pigmented tissue is ablated as an intended side effect.
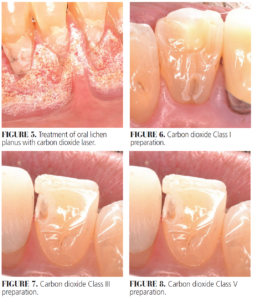
Low energy ablation therapy/de-epithelialization occurs at the epithelial surface and is not classified as ablation or PBM, rather the photothermal surface effect increases blood flow to the treated area resulting in faster healing. This can be accomplished with erbium and CO2 lasers and can be helpful in treatment of oral conditions such as lichen planus and spongiotic gingivitis (Figure 5).6
Snoring
Erbium and CO2 lasers can be used in a nonablative process to minimize snoring. Er:YAG lasers create subablative micropulses that initiate two processes.7 First, an indirect triggering effect by short duration heat shocking of the epithelium. Second is a direct slow thermal injury of the connective tissues. The result is neocollagenesis and the remodeling of collagen. This leads to firmer soft palate tissue and reduced vibrations. CO2 laser energy is applied to the soft palate, which creates cross-linking of collagen, causing it to contract. In its contracted form, collagen will reform stronger cross-linkages. Once treated, the soft palate is stiffer and less likely to vibrate.8
Photobiomodulation
PBM is used to reduce inflammation and promote healing.9 Its effectiveness depends on wavelength and energy density. Diodes and Nd:YAG are not absorbed in water; they penetrate deep into tissues to initiate biochemical responses in mitochondria. Optimal wavelengths are 660 to 1064 nm and dosages range from 4 to 8 J/cm2. PBM is used to stimulate healing for aphthous ulcers, temporomandibular disorders, extractions, soft tissue surgeries, and post-operative pain management following tooth extraction or implant placement. PBM can significantly reduce both healing time and amount of pain experienced.10
Cavity Preparations and debonding crowns
CO2 and Er:YAG lasers offer a highly effective option for a variety of hard tissue procedures including cavity preparations. Compared to rotary handpieces, lasers differ in three aspects. First, when using a laser, the practitioner relies on visual cues for cutting efficiency vs tactile sensations created by a bur. Distance to the target and hand speed are variables to master as these lasers operate in a noncontact mode approximately 10 mm above the tooth structure. Second, dental burs are usually end-cutting and side-cutting while lasers are end-cutting only. Third, these lasers are often able to perform most procedures, including class II cavity preparations without local anesthesia. This is especially advantageous for pediatric and highly anxious patients.
CO2 and erbium lasers are absorbed in water and hydroxyapatite. Er:YAG laser energy is almost 100x more absorbed in water than hydroxyapatite while CO2 laser energy is almost 100x more absorbed in hydroxyapatite than water. The result is that both lasers can manipulate tooth structure and bone; erbium by photoacoustic interaction, and CO2 by photothermal reaction.
Water spray is used with erbium lasers primarily to hydrate tooth structure ensuring efficient cutting while keeping the tooth cool even though the photoacoustic effect generates minimal heat. The photoacoustic interaction creates explosions in enamel and dentin ejecting tooth particles that create the preparation. The photothermal tissue interaction of CO2 with tooth structure ablates or vaporizes enamel and dentin creating the tooth preparation. Water spray is used to reduce heat and minimize thermal damage to pulpal tissues. Both lasers are effective in removing enamel and dentin but for quite varied reasons (Figures 6-8).
The removal of bonded crowns can be time consuming and requires multiple burs with rotary handpieces. Erbium laser energy is transmitted through ceramic materials and is absorbed in the cement layer beneath. Laser energy disrupts the chemical bond present between the tooth and the cement, and the cement and the crown. After 3 to 5 minutes when the laser energy is painted on all surfaces of the crown, a scaler may be used to dislodge the crown if needed. On rare occasions when the crown is not dislodged and when a rotary handpiece is needed, the bonded crowns are easily removed. Zirconia crowns with a higher yttria content are associated with a decreased crown retrieval time.11 Unlike erbium lasers, CO2 lasers are not able to debond crowns but can be used to section and remove a crown. Due to the lack of tactile sensation and the brightness of the ablation, rotary handpieces are considered more effective.
Endodontics
Erbium lasers are also useful in endodontic procedures because of their photoacoustic effects. Once the irrigating solution has been introduced to the canal system, these lasers may deploy two different techniques to assist endodontic procedures.
First, photon-induced photoacoustic streaming (PIPS) creates nonthermal photoacoustic waves within the cleaning and debriding solutions introduced in the canal, effectively cleaning canals and subcanals and freeing dentinal tubules of the smear layer.12 During obturation, sealer can reach portions of the canal anatomy that would be occluded without the use of erbium lasers. Second, Er:YAG lasers are used to improve debridement and disinfection creating synchronized pairs of ultra-short pulses, generating an accelerated collapse of laser-induced bubbles. This leads to enhanced shockwave emission even inside the narrowest root canals, known as shock wave-enhanced emission photoacoustic streaming (SWEEPS).12 PIPS is often compared to a single shotgun while SWEEPS is compared to an automatic gun.
Tooth Hypersensitivity
Diodes, Nd:YAG, erbium, and CO2 lasers can all address hypersensitivity. Hypotheses on the mechanisms of action, however, differ. Some surmise that protein on the surface of the tooth structure is coagulated without altering the surface of the tooth structure itself, indicating that lasers melt the surface of the tubules and create a frozen smear layer.13 Others suggest plasma inside the tooth structure precipitates, altering the activity of the nerve fiber.14 Laser power settings are generally 10x lower than surgical settings, which prevent irreversible damage to the tooth surfaces. In comparing the effectiveness of Nd:YAG laser vs diode, Nd:YAG was found to be better at long-term hypersensitivity reduction15 while other studies have shown that Er:YAG is effective at long-term dentin desensitizing.
Peri-Implantitis
Laser therapy should be considered in conjunction with mechanical debridement of the ailing implant. However, long-term data on using lasers of any variety to treat peri-implantitis are not available. Current treatment of peri-implantitis includes removing the inflamed/granulation tissue, removing diseased cortical bone, decontaminating the implant surface, and stimulating new bone growth. Use of diode or Nd:YAG lasers is contraindicated due to the risk of thermal damage to the implant surface and surrounding bone and should only be considered at the end of treatment to stimulate a fibrin clot.
Erbium and CO2 laser energy is reflected off dental implant surfaces and can be safely used to remove granulation tissue, decorticate the pocket, and debride and decontaminate the implant surface. CO2 lasers provide adequate water spray, minimizing photothermal effects and erbium lasers keeps bone hydrated. Photoacoustic effects associated with Er:YAG lasers have also been shown to clean biofilm from narrow geometries inaccessible to instrumentation on implant surfaces.16
Future Applications
New applications and wavelengths are constantly being evaluated for use in dentistry. A blue light diode laser (445 nm) is offers unique antimicrobial effects and the ability to use an uninitiated tip. The 445 nm wavelength has a much higher affinity for hemoglobins than 950 to 970 nm wavelengths and can operate at a fraction of the power output. The result is a clean fast cut with lower thermal collateral damage to surrounding tissues.17 Erbium lasers are used for surgical removal of bone during extractions followed by disinfection of the socket prior to grafting and/or implant placement. Outcomes are expected to be favorable vs nonlased sockets. Today PBM is used in orthodontics to accelerate tooth movement. Studies show dramatic decreases in overall treatment time. High-intensity laser therapy is also being evaluated for stimulation of healing and pain management.
References
- Wang HL, Avila-Ortiz G, Monje A, et al. AO/AAP consensus on prevention and management of peri-implant diseases and conditions: Summary report. J Periodontol. 2025;96:519-541.
- Schwarz F, Derks J, Monje A, Wang HL. Peri-implantitis. J Clin Periodontol. 2018;45(Suppl 20):S246-S266.
- Galarraga-Vinueza ME, Pagni S, Finkelman M, Schoenbaum T, Chambrone L. Prevalence, incidence, systemic, behavioral, and patient-related risk factors and indicators for peri-implant diseases: An AO/AAP systematic review and meta-analysis. J Periodontol. 2025;96:587-633.
- Kotsakis G, Olmedo D. Peri-implantitis is not periodontitis: Microbiome-biomaterial interactions. Periodontol 2000. 2021;86:231-240.
- Roccuzzo A, Imber JC, Marruganti C, Salvi GE, Ramieri G, Roccuzzo M. Clinical outcomes of dental implants in patients with and without history of periodontitis: A 20-year prospective study. J Clin Periodontol. 2022;49:1346-1356.
- Lin GH, Chan HL, Wang HL. The significance of keratinized mucosa on implant health: a systematic review. J Periodontol. 2013;84:1755-67.
- Lin GH, Lee E, Barootchi S, Rosen PS, Curtis D, Kan J, Wang HL. The influence of prosthetic designs on peri-implant bone loss: An AO/AAP systematic review and meta-analysis. J Periodontol. 2025;96:634-651.
- Guney Z, Altingoz SM, Has H, Serdar MA, Kurgan S. The impact of electronic cigarettes on peri-implant health: A systematic review and meta-analysis. J Dent. 2024;143:104883.
- Mojaver S, Zad A, Sarmiento H, Fiorellini JP. Efficacy of supportive peri-implant therapy in the management of peri-implant mucositis and peri-implantitis: A systematic review. J Am Dent Assoc. 2025;S0002-8177:00497-0.
- Ravidà A, Dias DR, Lemke R, Rosen PS, Bertolini MM. Efficacy of decontamination methods for biofilm removal from dental implant surfaces and reosseointegration: an AAP/AO systematicreview on peri-implant diseases and conditions. Int J Oral Maxillofac Implants. 2025;4:91-160.
- Maiorani C, Butera A, Pérez-Albacete Martínez C, et al. Effectiveness of erythritol-based air polishing and ultrasonic instrumentation with peek inserts in peri-implant maintenance: a randomized clinical trial including different prosthetic materials. Dent J (Basel). 2025;13:235.
- Tütüncüoğlu S, Cetinkaya BO, Pamuk F, et al. Clinical and biochemical evaluation of oral irrigation in patients with peri-implant mucositis: a randomized clinical trial. Clin Oral Investig. 2022;26:659-671.
- Maghsoudi P, Valkenburg C, Ter Gunne LP, van der Weijden FGA. Retrospective evaluation of peri-implant maintenance in patients with implant-supported fixed prostheses. Int J Dent. 2025;2025:9920951.
- Araújo TG, Moreira CS, Neme RA, Luan H, Bertolini M. Long-term implant maintenance: a systematic review of home and professional care strategies in supportive implant therapy. Braz Dent J. 2024;35:e246178.
- Salvi GE, Bosshardt DD, Lang NP, et al. Temporal sequence of hard and soft tissue healing around titanium dental implants. Periodontol 2000. 2015;68:135-52.
- Monje A, Barootchi S, Rosen PS, Wang HL. Surgical- and implant-related factors and onset/progression of peri-implant diseases: An AO/AAP systematic review. J Periodontol. 2025;96:542-561.
From Decisions in Dentistry. February/March 2026;12(1):20-23.