Managing Oral Mucositis
An in-depth look at the causes, complications, and multidisciplinary approaches to prevent and manage oral mucositis in patients undergoing chemotherapy and radiation therapy.
This course was published in the July/August 2025 issue and expires August 2028. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
AGD Subject Code: 730
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Identify the pathophysiology and clinical manifestations of oral mucositis associated with chemotherapy and radiation therapy in patients with head and neck cancer.
- Describe current evidence-based prevention and management strategies for oral mucositis.
- Discuss the role of oral health professionals in educating, supporting, and implementing care plans to improve quality of life and treatment outcomes for patients at risk of or experiencing oral mucositis.
Oral mucositis is a common and debilitating side effect of head and neck cancer (HNC) treatment, including chemotherapy and radiation therapy. Involving inflammation and ulceration of the mucous membranes in the mouth, oral mucositis causes pain and difficulty swallowing, as well as raises the risk of infection. Patients often experience significant discomfort, which can affect their ability to eat, speak, and maintain oral hygiene. Managing this severe oral side effect typically involves a combination of preventive measures and addressing specific symptoms. Symptoms are treated with a combination of pain relief, oral hygiene, and, in severe cases, the use of medications to promote healing. Preventive strategies include cryotherapy and the use of protective mouthrinses.
Mechanism of Radiation Therapy
A cornerstone of HSC treatment, radiation therapy uses ionizing radiation to target and destroy cancer cells by damaging the cell DNA. This process prevents the cells from dividing and growing. While radiation affects both cancerous and normal cells, the goal is to maximize the dose to cancer cells while minimizing exposure to healthy tissues.1 Healthy tissues near the tumor can receive high radiation doses, resulting in toxicity classified as acute (within 90 days) or late (after 90 days).2
Acute toxicity commonly occurs with both external beam radiotherapy (EBRT) and chemotherapy, often leading to synergistic effects, including mucositis, dysphagia, and loss of taste. Late toxicities involve osteoradionecrosis, xerostomia, fibrosis, thyroid dysfunction, and trismus.3
Oral Complications from Radiation
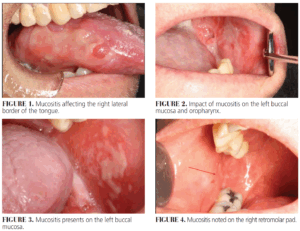
Acute toxicities caused by radiation include oral mucositis, dsyphagia, dysguesia, and xerostomia. Oral mucositis — inflammation of the oral and oropharyngeal mucosa — typically appears within the first 2 to 3 weeks of treatment, causing significant discomfort and nutritional challenges. Nearly all patients undergoing chemoradiation experience mucositis, impacting their quality of life and treatment success (Figures 1-4).3,4
Common among older adults, dysphagia, or difficulty swallowing, results from damage to the upper aerodigestive tract tissues.5 Dysphagia prevalence increases during treatment and often persists post-treatment, affecting 45% to 75% of HNC survivors. The rising incidence of HNC, particularly among patients treated for human papillomavirus-associated oropharyngeal cancers, suggests an increasing number of survivors with dysphagia.6
Dysgeusia, or taste alteration, affects about 76% of patients undergoing head and neck chemoradiation therapy. Taste changes worsen significantly between the 4th and 8th week of treatment.7
Xerostomia impacts more than 90% of patients undergoing radiotherapy, causing problems with chewing, swallowing, speech, and oral hygiene. Xerostomia typically begins about 4 weeks after starting radiation therapy and can take up to 18 months to improve. Preventive strategies, such as submandibular gland transfer and shielding, can help reduce xerostomia by preserving salivary function.8
Late toxicities of radiation therapy include trismus and osteoradionecrosis. Trismus is limiting of the mouth opening due to radiation affecting the temporomandibular joint or masticatory muscles, which may cause muscle fibrosis and scarring. Trismus can interfere with nutrition, speech, and dental care; it may persist for life. Therapeutic interventions include pentoxifylline, botulinum toxin, and exercises.3
Osteoradionecrosis is a severe condition in which irradiated tissue breaks down and fails to repair, leading to exposed and necrotic bone. The mandibular bone is particularly susceptible, and the risk increases with radiation doses above 60 Gy. Osteoradionecrosis occurs in approximately 5% to 15% of patients who have undergone radiotherapy. Pre-radiation dental management is crucial to minimize the need for tooth extractions afterward. Treatment often involves surgical resection and vascular graft reconstruction.3
Other Types of Radiation Therapy
A versatile and effective treatment for advanced localized cancers, EBRT works by inducing DNA damage, cell cycle arrest, and apoptosis in cancer cells, preventing proliferation and leading to cell death. Normal cells, although exposed to some radiation, can repair damage faster than cancer cells, minimizing collateral harm. Advances in imaging, dose delivery, and specialized techniques have improved treatment precision and outcomes.9
Brachytherapy plays a crucial role in HNC management due to its ability to deliver focused radiation while safeguarding nearby structures. Brachytherapy involves placing encapsulated radionuclides directly within or near a tumor, offering highly localized radiation treatment with outcomes comparable to surgery for T1 to T2 tumors.10,11
Chemotherapy
Chemotherapy aims to inhibit tumor growth, proliferation, and metastasis by interfering with DNA, RNA, or protein synthesis, often triggering cell death through apoptosis. However, its distribution and impact on normal cells result in significant side effects.
Traditional chemotherapy is most effective during specific cell cycle phases, such as the S phase (DNA synthesis) and M phase (mitosis), with agents like vinca alkaloids and taxanes targeting mitotic spindle formation. Multiple treatment cycles may be necessary due to delayed cell death and the need for cumulative responses. Chemotherapy agents can be administered through various routes, including oral, intravenous, subcutaneous, intramuscular, and intrathecal. Intravenous is the most common due to its 100% absorption rate.
The incidence and severity of mucositis depend on factors such as the chemotherapy regimen, dosage, and treatment schedule.12 Chemotherapeutic agents are classified by their mechanisms of action, such as alkylating agents inhibiting DNA replication and transcription, and antimetabolites blocking DNA replication.13 Multidrug regimens are associated with decreased salivary flow and altered salivary composition. This hyposalivation raises the risk of oral mucositis. Research involving animal models shows that 5-fluorouracil induces oxidative stress, apoptosis, and atrophy in salivary gland acinar cells due to inflammation and tissue damage.14 These effects weaken the glands’ protective functions, increasing the severity of oral mucositis. Reduced salivary flow also slows mucosal ulcer healing due to diminished salivary clearance and antimicrobial activity.14
Oral Mucositis Prevention and Management
Several therapeutic options are available to prevent oral mucositis. Maintaining good oral hygiene is key not only for preventing infections but also for overall health.15 Poor oral hygiene can lead to periodontal diseases, which may increase the risk of infections, especially among individuals with compromised immune systems or those with mucosal ulcerations. Keeping the mouth clean helps minimize harmful bacteria, reducing the likelihood of sepsis and promoting faster healing of any existing sores. Regular brushing, flossing, and dental check-ups play an essential role in this preventive care.16
Oral cryotherapy is quite effective in managing the side effects of chemotherapy. The introduction of local vasoconstriction helps reduce blood flow to the mucosal tissues, which can lower the exposure of these tissues to the harmful effects of chemotherapy drugs.16 Vasoconstriction can minimize the severity of mucositis, a common and painful side effect of cancer treatment. Additionally, cryotherapy may provide pain relief and promote healing in the oral mucosa.
Ice chips, ice cubes, and ice lollipops are common forms of oral cryotherapy. Patients can suck on these to cool the oral cavity, which may mitigate the risk of mucositis during chemotherapy. The cold temperature not only promotes local vasoconstriction but can also provide soothing relief for any discomfort. It’s a simple yet effective way to enhance oral care during treatment. Overall, cryotherapy is a helpful adjunctive therapy for patients undergoing chemotherapy.
Several coating agents have been developed to create protective barriers on the oral mucosa, helping to reduce irritation from oral mucositis. These agents often consist of bioadhesive gels or films that adhere to the mucosal surfaces, providing a shield against irritants and reducing pain.17 Topical agents can also promote healing by maintaining moisture and creating a favorable environment for tissue repair. These formulations can be especially beneficial for patients undergoing chemotherapy or radiation therapy.17
Managing oral mucositis requires a comprehensive, multidisciplinary approach aimed at reducing its severity and associated pain. Optimizing oral health before treatment and administering analgesics for pain control are key. Numerous clinical trials have investigated palliative treatments to prevent and manage oral mucositis, underscoring the continued search for effective therapeutic interventions. This integrated approach ensures better patient outcomes by addressing both the cause and symptoms of oral mucositis.14,18
Inflammation plays a critical role in the development of oral mucositis caused by radiotherapy and chemotherapy, with pro-inflammatory cytokines contributing significantly to its pathogenesis. Research indicates that benzydamine effectively prevents oral mucositis related to radiotherapy and chemoradiotherapy, demonstrating statistically significant results.19
Microbial colonization is a key factor in sustaining the inflammatory response in oral mucositis, making antimicrobial agents a promising preventive measure. Additionally, herbal medicines have garnered attention due to their affordability, availability, and fewer side effects compared to synthetic drugs. Herbal agents, such as curcumin exhibit antioxidant, anti-inflammatory, analgesic, antifungal, antiseptic, and anticancer properties, with studies confirming their effectiveness in reducing oral mucositis severity.19 Moreover, the United States Food and Drug Administration has recently approved keratinocyte growth factor to treat mucositis, offering new therapeutic avenues for cancer patients undergoing aggressive treatments.18,19
Photo biomodulation (PBM) is a form of low-level laser therapy that uses specific wavelengths of light to stimulate biological responses in tissues. This noninvasive treatment can be particularly beneficial in managing oral mucositis and promoting healing.
Clinical studies have indeed shown that low-level laser therapy, or photo biomodulation, can effectively reduce the severity of oral mucositis in patients undergoing chemotherapy and radiation therapy. Many studies indicate that low-level laser therapy can significantly decrease the severity of mucositis symptoms, leading to less pain and discomfort.15,20
Patients receiving low-level laser therapy often experience a shorter duration of mucositis, allowing for a better quality of life during treatment. Low-level laser therapy promotes faster healing of mucosal tissues, potentially preventing complications associated with severe mucositis. Additionally, it can help reduce the need for analgesics and other pain management strategies, leading to more comfortable treatment experiences.
Low-level laser therapy plays a significant role in promoting tissue repair by enhancing mitochondrial function, which in turn accelerates the healing process in oral tissues. In addition to its regenerative benefits, low-level laser therapy helps modulate inflammatory responses, leading to reduced swelling and improved recovery outcomes.
In terms of practical application, low-level laser therapy is generally most effective when initiated early in the treatment process or at the first signs of mucositis. Various protocols are available, with differences in light wavelength, dosage, and treatment frequency. These protocols can be customized to suit the specific needs of each patient, allowing for a more personalized and effective approach to care. In many states, dental hygienists are allowed to provide low-level laser therapy. This enables them to offer valuable support in managing conditions like oral mucositis. In dental practices where dental hygienists provide low-level laser therapy, the utilization expands the scope of practice and patient care, supporting the multidisciplinary approach to managing oral health during cancer treatment.
Recent studies indicate that nutritional therapy using an oral amino acid-rich enteral diet can significantly enhance healing through improved protein synthesis.21 This is largely attributed to the diet’s composition, which is high in essential amino acids. These amino acids are crucial for various physiological processes, including tissue repair and immune function.
Toyomasu et al22 conducted a prospective, randomized clinical trial of patients who underwent chemotherapy for gastric cancer. The main objective was to evaluate the efficacy of an amino acid-rich product in reducing oral mucositis and minimizing body weight loss in patients undergoing chemotherapy for gastric cancer. The study included two groups of participants: a test group of 11 patients who consumed an amino acid-rich product, and a control group of 11 patients who received standard nutritional care. The primary outcome measured was the incidence of oral mucositis, while the secondary outcome focused on the median body weight loss during treatment.
The results showed that the incidence of oral mucositis was significantly lower in the treatment group at 9.1%, compared to 27.3% in the control group. Additionally, the treatment group experienced a significantly smaller median body weight loss than the control group, with a P value of .015 indicating statistical significance. These findings suggest that consuming amino acid-rich products may effectively reduce the occurrence of oral mucositis and help minimize weight loss in patients undergoing chemotherapy for gastric cancer.
Education and Support
Educating patients about the signs and symptoms of oral mucositis before starting chemotherapy or radiation therapy is crucial for effective early intervention. Providing patients with written materials, oral care guidelines, and information about supportive products can further empower them to take charge of their oral health during the treatment process. The proactive educational approach is key to improving outcomes and overall patient experience during cancer therapy.16
Conclusion
Preventive strategies may vary based on individual risk factors and treatment protocols; therefore, patients must work closely with their healthcare team to find the most suitable approach. Oral health professionals are uniquely positioned to positively impact patient well-being through the use of measures to decrease symptoms of oral mucositis.
References
- Baskar R, Lee KA, Yeo R, Yeoh K. Cancer and radiation therapy: Current advances and future directions. Int J Med Sci. 2012;9:193–199.
- Singh B, Singh G, Oinam AS, et al. Radiobiological modeling of radiation-induced acute mucosal toxicity (oral mucositis and pharyngeal mucositis): A single-institutional study of head-and-neck carcinoma. J Cancer Res Ther. 2021;19(Suppl 1):S380.
- Alfouzan AF. Radiation therapy in head and neck cancer. Saudi Med J. 2021;42:247.
- Goel G, Alice KP, Negi P, Aggarwal N. Factors influencing radiation induced oral mucositis in head and neck cancer. J Cancer Res Ther. 2024;20:1564–1569.
- Sroussi HY, Epstein JB, Bensadoun R, et al. Common oral complications of head and neck cancer radiation therapy: Mucositis, infections, saliva change, fibrosis, sensory dysfunctions, dental caries, periodontal disease, and osteoradionecrosis. Cancer Med. 2017;6:2918–2931.
- Kuhn MA, Gillespie MB, Ishman SL, et al. Expert consensus statement: Management of dysphagia in head and neck cancer patients. Otolaryngol Head Neck Surg. 2023;168:571–592.
- Togni L, Mascitti M, Vignigni A, et al. Treatment-related dysgeusia in oral and oropharyngeal cancer: A comprehensive review. Nutrients. 2021;13:3325.
- Brook I. Early side effects of radiation treatment for head and neck cancer. Cancer/Radiothérapie. 2021;25:507–513.
- Koka K, Verma A, Dwarakanath BS, Papineni RV. Technological advancements in external beam radiation therapy (EBRT): An indispensable tool for cancer treatment. Cancer Manag Res. 2022;14:1421–1429.
- Guinot JL, Bacorro W, Budrukkar A, et al. GEC-ESTRO recommendations for head & neck cancer brachytherapy (interventional radiotherapy): 2nd update with focus on HDR and PDR. Radiother Oncol. 2024;201:110533.
- Wierzbicka M, Bartochowska A, Strnad V, et al. The role of brachytherapy in the treatment of squamous cell carcinoma of the head and neck. Eur Arch Otorhinolaryngol. 2016;273:269–276.
- Pulito C, Cristaudo A, Porta CL, et al. Oral mucositis: the hidden side of cancer therapy. J Exp Clin Cancer Res. 2020;39:210.
- Amjad MT, Chidharla A, Kasi A. Cancer Chemotherapy. Treasure Island, Florida: StatPearls Publishing; 2024.
- Shetty SS, Maruthi M, Dhara V, et al. Oral mucositis: Current knowledge and future directions. Dis Mon. 2021;68(5):101300.
- Pulito C, Cristaudo A, La Porta C, et al. Oral mucositis: the hidden side of cancer therapy. J Exp Clin Cancer Res. 2020;39:210.
- Bell A, Kasi A. Oral Mucositis. Treasure Island, Florida: StatPearls Publishing; 2024.
- Ueno T, Yatsuoka W, Ishiki H, Miyano K, Uezono Y. Effects of an oral mucosa protective formulation on chemotherapy- and/or radiotherapy-induced oral mucositis: a prospective study. BMC Cancer. 2022;22:90.
- Blakaj A, Bonomi M, Gamez ME, Blakaj DM. Oral mucositis in head and neck cancer: Evidence-based management and review of clinical trial data. Oral Oncol. 2019;95:29-34.
- Colella G, Boschetti CE, Vitagliano R, et al. Interventions for the prevention of oral mucositis in patients receiving cancer treatment: evidence from randomized controlled trials. Current Oncol. 2023;30:967-980.
- Elad S, Cheng KKF, Lalla RV, et al. MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer. 2020;126:4423-4431.
- Tanaka Y, Shimokawa T, Harada K, Kazuhiro Yoshida K. Effectiveness of elemental diets to prevent oral mucositis associated with cancer therapy: A meta-analysis. Clin Nutr ESPEN. 2022;49:172-180.
- Toyomasu Y, Mochiki E, Yanai M, et al. A prospective pilot study of an elemental nutritional supplement for prevention of oral mucositis during S-1 adjuvant chemotherapy for gastric cancer. Surg Oncol. 2019;29:97e101.
From Decisions in Dentistry. July/August 2025;11(4):40-45.