Immune Response in Patients With Type 2 Diabetes or Human Immunodeficiency Virus
Given the number of patients presenting with these maladies, understanding key differences in immune response will help guide appropriate oral care.
This course was published in the June 2017 issue and expires June 2020. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Discuss how the innate and adaptive immune systems work.
- Identify dental treatment considerations for patients with type 2 diabetes.
- Explain how human immunodeficiency virus (HIV) affects the adaptive immune system.
- Detail oral care considerations for patients with HIV infection.
Type 2 diabetes mellitus (T2DM), human immunodeficiency virus and acquired immunodeficiency syndrome (HIV/AIDS) infection are common diseases affecting the human immune system. It is estimated that more than 29 million Americans have diabetes, with T2DM representing 90% to 95% of diagnosed cases.1 More than 1.2 million adolescent and adult Americans are infected with HIV/AIDS, with approximately 50,000 new cases each year. The U.S. Centers for Disease Control and Prevention reports that among these individuals, 13% are unaware they have been infected.2 When caring for these patients, oral health professionals may ask how the associated attacks on the immune system differ. Are the medical and dental risks similar? And what treatment modifications should be considered? Another pressing question is whether dentists have a role in the diagnosis of these two illnesses. This article provides an overview of these considerations.
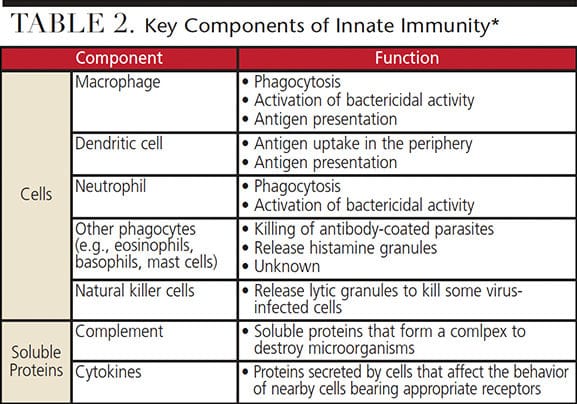
The human body is armed with two major immune systems, innate and adaptive (Table 1). The innate immune system is present at birth. The body uses this system immediately, or within hours after exposure to almost any microbe, to prevent infection (Figure 1 and Table 2). Unlike the adaptive immune system, the innate system does not recognize every possible invader. It is designed to recognize certain essential molecules that are shared by a number of related pathogenic microbes that are not associated with the cell design in mammals.3 The innate immune system response does not improve with repeated exposure to a given pathogen.3 This immune response system appears to be affected by diabetes, resulting in periodontal disease of increased virulence.1
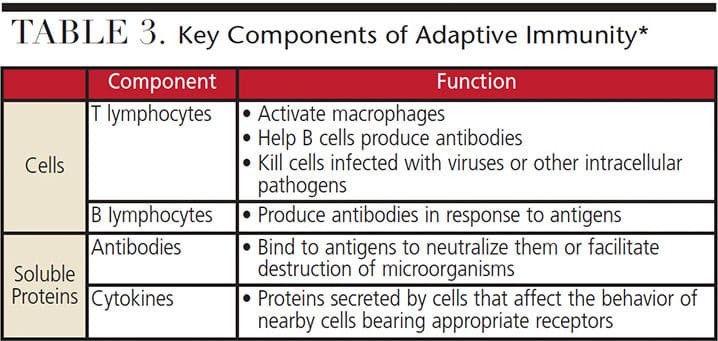
As its name indicates, the adaptive immune system develops throughout life (Table 3). This system provides the ability to react to specific foreign invaders, also known as antigens (antibody generator). It does so by binding them with B-cell (B-lymphocytes) secreted antibodies. This antibody binding marks the invading pathogen, making it easier for phagocytic cells of the innate immune system to ingest them.3
Additional groups in the cell-mediated adaptive immune response are activated T-cell (T-lymphocytes) and T-helper cells. T-cells react against a foreign antigen. To ensure the effectiveness of the adaptive immune system, the body develops massive numbers of T-cell and B-cell clones that allow it to recognize and react to antigens.3 This system improves its response with repeated exposures to a given stimuli or antigen. The ability to distinguish foreign from self is a fundamental feature of the adaptive immune system, as failure to recognize self can lead to destruction of the host’s own molecules — as is seen in Type 1 diabetes mellitus when the beta cells of the pancreas are destroyed.3
DIABETES AND THE INNATE IMMUNE SYSTEM
T2DM is a metabolic disorder that can bring about complications, including dysregulation of the innate immune system and an associated increase in inflammatory response. This dysregulation leads to the excessive formation and accumulation of advanced glycation end products (AGEs) in tissues inducing oxidative stress, and is thought to play a role in periodontal inflammation in the patients with diabetes.4,5 Research suggests the presence of bacteria in the periodontal pocket triggers the innate immune system to send neutrophils to phagocytize the invading bacteria and initiate a rapid inflammatory response.5
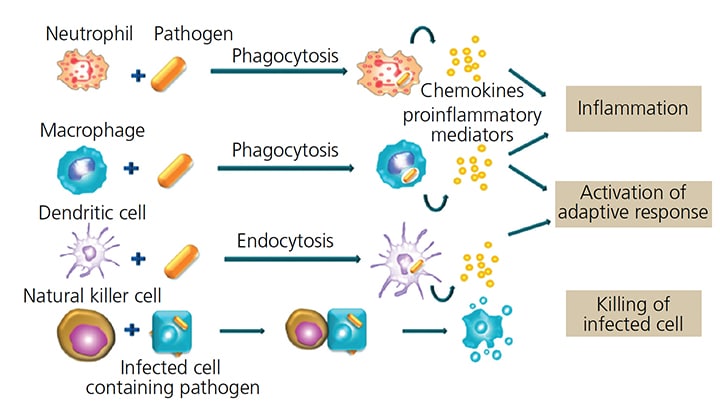
When microorganisms overcome the innate system’s first line of defense, a second line is activated that includes macrophages, lymphocytes and cytokines.5 It appears that AGEs may be deposited on polymorphonuclear cells that produce a hyperinflammatory state, which amplifies the body’s response to cytokines released upon contact with an antigen.5,6 These activated neu-trophils show a heightened response upon contact with the lipopolysaccharide of gram-negative bacteria, such as the periodontal pathogen Porphyromonas gingivalis that is found in subgingival biofilm.7 This heightened neutrophil behavior consequently triggers an exaggerated inflammatory cascade, which increases the destruction of periodontal connective tissue.7
ORAL HEALTH CONSIDERATIONS FOR PATIENTS WITH type 2 DIABETES
Periodontal disease is one of many known complications of T2DM.5,6 The effect of diabetes on the innate immune system is thought to play a major role in the progression of periodontal disease, in part through the production of proinflammatory cytokines.8 The process by which diabetes contributes to this increased virulence of periodontal disease is complex and continues to be investigated. Potential complications resulting from T2DM include enhanced gingival bleeding, increased loss of attachment, greater pocket depth formation, increased loss of alveolar bone and poor wound healing.7 The presence of opportunistic infections, such as oral candidiasis, may also be a manifestation of systemic immunosuppression associated with diabetes.9
Oral health professionals should be aware of these effects on nonsurgical and surgical periodontal treatment modalities. Although uncontrolled diabetes is a contraindication to elective procedures, even with good glycemic control, clinicians can expect delayed wound healing in these patients. This is due to diabetes-induced hypoxia, dysfunction in fibroblasts and dermal cells, impaired angiogenesis and other factors — including damage from AGEs and decreased host immune resistance.8
If periodontal or other oral surgery is planned, good glycemic control will support healing and a positive outcome. Various tests are available to monitor glycemic control. The HbA1c test for glycated hemoglobin is the best indicator of long-term glycemic control5 (an A1c or HbA1c reading of < 6.5% is ideal; retest every three to six months). Response to initial nonsurgical scaling and root planing may help predict outcomes and prognosis of subsequent treatment, including healing following periodontal surgery.10 Ship9 advises that, “Managing patients with diabetes requires more rigorous follow-up, more aggressive interventional therapy rather than observation, regular communication with physicians, and greater attention to prevention. Patients with diabetes, particularly those with a history of poor glycemic control and oral infections, require more frequent recall visits and fastidious attention to acute oral infections.”
HUMAN IMMUNODEFICIENCY VIRUS
The classic effect of HIV infection is on the adaptive immune system and is more specific than the generalized disruption of the innate immune system seen with T2DM. HIV-related disruption affects the T-lymphocyte branch of the immune system and the destruction of CD4+ T-helper cells, in particular.2 The terms “T-cell” and “CD4 cell” are used interchangeably when discussing HIV. Both refer to the same type of cell, and, while there are many different types of T-cells, these particular cells have a specific receptor called the CD4 receptor site. HIV uses this receptor to latch on to the T-cell, making it a prime target for infection (Figure 2).2
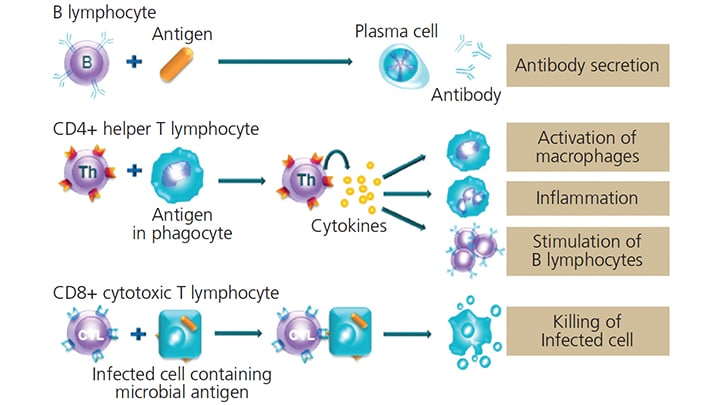
When infection occurs, the adaptive immune system generates a population of “memory” B-cells that quickly mount an antibody response if the invading antigen is encountered again.3 HIV infection appears to create an unusual population of exhausted and unresponsive B-memory cells, which diminish the immune response. It is thought the production of unresponsive B-memory cells allows the HIV to reassert itself, leading to increasing numbers of HIV. Thus, the infection is allowed to continue, potentially overwhelming the body’s ability to respond appropriately.11
CONSIDERATIONS FOR PATIENTS WITH HUMAN IMMUNODEFICIENCY VIRUS
Patients with HIV may present with conditions that include acute necrotizing ulcerative gingivitis or periodontitis, oral candidiasis, herpes simplex and zoster viruses, recurrent apthous ulcers and chronic periodontitis.12 Research has shown that the degree of attachment loss is greater in the HIV-infected patient with preexisting chronic adult periodontal disease, particularly when CD4 counts drop below 200 cells/mm3.13
Since the introduction of highly active antiretroviral therapy (HAART) in 1995, the course of HIV disease has been modified to a manageable chronic illness.14 Use of HAART has become the standard of care in the treatment of patients with HIV, and appears to have decreased the prevalence, severity and course of associated periodontal disease.14 This therapy generally increases the CD4 cell count above 200 cells/mm3 and is thought to decrease inflammation and immune activation, while producing fewer side effects than is seen with previous antiretroviral therapy.
The oral cavity appears to be a site of HIV pathogenesis and a potential reservoir for the disease.14 HIV RNA and DNA forms are present in saliva, as well as in gingival crevicular fluid.14 Oral epithelial cells are susceptible to either cell-free or cell-associated HIV infection.14 This reservoir may lead to reactivation of the virus, even in those undergoing HAART therapy.14 Researchers are investigating the potential role of periodontitis, especially untreated conditions, as a global oral infection that potentially contributes to HIV renewed outbreak.14
Although there is still concern that periodontal procedures, including scaling and root planing, may cause systemic bacteremia and subsequent systemic infections, evidence supports the assertion that bacteremia induced in this way returns to normal relatively quickly in patients with a CD4 count > 200 cells/mm3.15 In patients with HIV, it must also be acknowledged that bacteremia may occur not only as a result of professional dental manipulation, but also from routine daily activities, such as eating, toothbrushing or other oral self-care procedures. Good oral hygiene and gingival health are associated with reduced risk of bacteremia, making continuing care especially important in this patient population.16,17
While HIV infection may initially lead to a particular depression of the adaptive immune system, with a subsequent increase in opportunistic infections, it does not appear to alter the body’s overall ability to heal — as occurs in patients with T2DM. Research and review of treatment have demonstrated that HIV-positive patients with CD4 counts > 200 cells/mm3 generally have normal healing times following periodontal surgery (including implant placement),18 while those with a CD4 count < 200 cells/mm3 generally experience a slightly higher failure rate than patients without HIV.19
CONCLUSION
Dental teams must remain vigilant about the clinical considerations presented by patients with T2DM or HIV infection. Through intraoral examination and early, or refractory periodontal treatment failures, oral health professionals may be the first to recognize possible systemic pathology. With diabetes, a patient’s health history may present clues indicative of undiagnosed disease. Additionally, poor response to periodontal treatment, even in the face of optimal self-care, may raise a practitioner’s suspicions.
Simple in-office testing methods for blood sugar levels are available and can easily be utilized for screening of prediabetes and T2DM.20 In-office testing for HIV infection in the dental setting — though available — may be problematic due to the rate of false positive results.21 Referral to a physician for testing and potential diagnosis of T2DM or HIV should be the first line of care for patients not under routine medical care.
All possible risk factors must be weighed when diagnosing and treating individual patients. Periodontal treatment plans created for patients with T2DM or HIV infection should be tailored based on current medical and social history, appropriate lab test results, diagnosis of oral findings, and consideration of current evidence. Knowledge of the patients’ medical status will help ensure the safest, most efficient and effective dental care possible.
REFERENCES
- U.S. Centers for Disease Control and Prevention. Diabetes: Working to Reverse the U.S. Epidemic, At a Glance 2016. Available at: cdc.gov/chronicdisease/resources/publications/aag/diabetes.htm. Accessed May 5, 2017.
- AIDS.gov. HIV in the United States: At a Glance. Available at: aids.gov/hiv-aids-basics/hiv-aids-101/statistics. Accessed May 5, 2017.
- Alberts B, Johnson A, Lewis J, Raff M, Roberts K, and Walters P. Molecular Biology of the Cell. 4th Ed. New York and London: Garland Science; 2002.
- Virella G, Lopes-Virella M. The role of the immune system in the pathogenesis of diabetic complications. Front Endocrinol (Lausanne).2014;5:126.
- Bascones-Martínez A, González-Febles J, Sanz-Esporrín J. Diabetes and periodontal disease. Review of the literature. Am J Dent. 2014;27:63–67.
- Chapple IL, Genco R, working group 2 of the joint EFP/AAP workshop. Diabetes and periodontal diseases: consensus report of the Joint EFP/AAP Workshop on Periodontitis and Systemic Diseases. J Periodontol. 2013;84(4 Suppl):S106–12.
- Bascones-Martinez A, Matesanz-Perez P, Escribano-Bermejo M, González-Moles MÁ, Bascones-Ilundain J, Meurman JH. Periodontal disease and diabetics-review of the literature. Med Oral Patol Oral Cir Bucal. 2011;16:e722–e729.
- Guo S, DiPietro LA. Factors affecting wound healing. J Dent Res. 2010;89:219–229.
- Ship JA. Diabetes and oral health: an overview. J Am Dent Assoc. 2003;134(suppl):4S–10S.
- Lalla E, Hsu WC, Lamster IB. Dental and Medical Comanagement of Patients with Diabetes. In: Genco RJ, William RC, eds. Periodontal Disease and Overall Health: A Clinician’s Guide. Yardley, Penn: Professional Audience Communications Inc; 2010:224.
- Kardava L, Moir S, Wang W, et al. Attenuation of HIV-associated human B cell exhaustion by siRNA downregulation of inhibitory receptors. J Clin Invest. 2011;121:2614–2624.
- Vernon LT, Demko CA, Babineau DC, et al. Effect of Nadir CD4+ T cell count on clinical measures of periodontal disease in HIV+ adults before and during immune reconstitution on HAART. PLoS One. 2013;8:e76986.
- Stanford TW, Rees TD. Acquired immune suppression and other risk factors/indicators for periodontal disease progression. Periodont 2000. 2003;32:118–135.
- González OA, Ebersole JL, Huang CB.Oral infectious diseases: a potential risk factor for HIV virus recrudescence? Oral Dis. 2009;15:313–327.
- Lucartorto FM, Franker CIC, Maza J. Post scaling bacteremia in HIV-associated gingivitis and periodontitis. Oral Surg Oral Med Oral Pathol.1992;73:550–554.
- Lockhart PB, Brennan MT, Thornhill M, et al. Poor oral hygiene as a risk factor for infective endocarditis–related bacteremia. J Am Dent Assoc. 2009;140:1238–1244.
- Siberry, GK, Abzug MJ, Nachman S, et al. Guidelines for the prevention and treatment of opportunistic Infections in HIV-exposed and HIV-infected children: recommendations from the National Institutes of Health, Centers for Disease Control and Prevention, the HIV Medicine Association of the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the American Academy of Pediatrics. Pediatr Infect Dis J. 2013;32(suppl 2):i–KK4.
- Kolhatkar S, Khalid S, Rolecki A, Bhola M, Winkler JR. Immediate dental Implant placement in HIV-positive patients receiving highly active antiretroviral therapy: a report of two cases and a review of the literature of implants placed in HIV-positive individuals. J Periodontol. 2011;82:505–511.
- May MC, Andrews PN, Daher S, Reebye UN. Prospective cohort study of dental implant success rate in patients with AIDS. Int J Implant Dent. 2016;2(1):20.
- Herman WH, Taylor GW, Jacobson JJ, Burke R, Brown MB. Screeing for prediabetes and type 2 diabetes in dental offices. J Public Health Dent. 2015;75:175–182.
- AIDS.gov. HIV Test Types. Available at: aids.gov/hiv-aids-basics/prevention/hiv-testing/hiv-test-types/. Accessed May 5, 2017.
Featured Image by LUISMMOLINA/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. June 2017;3(6):32—35.