Saliva, Remineralization And Dental Caries
Along with preventive therapies, optimal salivary action can promote remineralization and reverse lesion development in patients with dental caries.
This course was published in the April 2019 issue and expires April 2022. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
EDUCATIONAL OBJECTIVES
After reading this course, the participant should be able to:
- Describe the cyclical process of tooth demineralization and remineralization.
- Explain saliva’s role in caries risk assessment and demineralization/remineralization.
- Discuss clinical strategies and tools for assessing and managing caries risk, and assisting remineralization of tooth structure.
The development of dental caries is a dynamic, multifactorial process. The constant cycle of tooth demineralization/remineralization can be positively or negatively impacted by a variety of oral and systemic factors. Assessing caries risk status and identifying appropriate remineralization therapies for high-risk individuals are key steps in improving patients’ oral health.
Considered a critical biological and protective factor in the remineralization of enamel, saliva is an important component in caries risk assessment and when selecting remineralization therapies.1 Saliva’s buffering capacity and production are directly related to the rate and extent of tooth demineralization.2 Saliva can neutralize acids, form a protective membrane on tooth surfaces, and enhance remineralization by providing calcium, phosphate and fluoride to enamel and dentin.3 Salivary pH levels directly affect remineralization through the amount of calcium and phosphate ions available to the enamel in times of acidic challenge.4 Saliva can act as a replenishing source and inhibit tooth demineralization during periods of low pH, while promoting tooth remineralization when the pH returns to a neutral state.5 Systemic conditions, hereditary disorders, a variety of medications, and other medical interventions can negatively affect salivary production, buffering potential, and the amount of calcium and phosphate available for remineralization. As such, evaluating a patient’s salivary flow should be a regular component of caries risk assessment.
Demineralization is the process of removing mineral ions from hydroxyapatite crystals of hard tissues, such as enamel, and, when unchecked, can lead to dental caries.5–7 Demineralization/remineralization is a continuous, cyclical process,5,7 and the former takes place on the tooth surface when biofilm — consisting of bacterial plaque and the pellicle — is present on enamel and dentin.8 When fermentable carbohydrates are ingested, Lactobacillus and Streptococcus bacteria in the biofilm metabolize the carbohydrates and produce acid.7 This acid diffuses across the tooth surfaces, dissolving minerals in the enamel and dentin.7
Although demineralization results in the loss of mineral ions, it can be reversed during remineralization. Both processes occur on the tooth surface, however, a considerable number of mineral ions must be lost from hydroxyapatite before cavitation occurs.5 The extent of demineralization and remineralization depends on several factors, including the amount of calcium and phosphate available, as well as salivary pH levels. Individuals with reduced salivary flow tend to have more acidic saliva and biofilm, raising the risk for continued demineralization and eventual lesion development.4,9
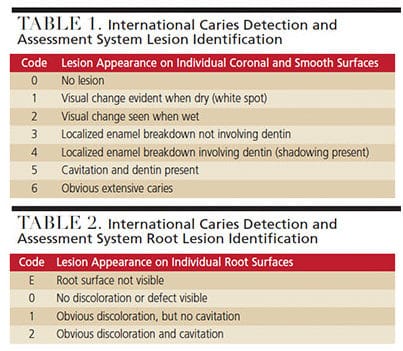
Patients with reduced salivary flow and/or compromised buffering potential need assistance with the remineralization cycle. While it is important to prevent early lesions, identifying dental caries in the early stages of the demineralization process — while the condition is still reversible — is critical. Oral health professionals can intervene with preventive remineralization therapies before the process becomes irreversible. The International Caries Detection and Assessment System (ICDAS) is a tool used to visually assess the development of early caries lesions on coronal, smooth and root surfaces (Table 1 and Table 2).8,10
HYPOSALIVATION
Hyposalivation and/or xerostomia is a decrease in salivary flow and is characterized by oral dryness.11 It can be caused by medications, autoimmune diseases (such as Sjögren’s syndrome), radiotherapy/chemotherapy and hormone disorders.11 Approximately 30% of patients between the ages of 20 and 69 have xerostomia,12 and medications are the most common cause.13 As life expectancy grows, the prevalence of xerostomia increases due to the number of medications prescribed for various health conditions.13 A review by Smith and Burtner14 revealed xerostomia was the most common side effect (80.5%) of 200 of the most frequently prescribed medications in the United States. Patients who experience diminished salivary production are at an increased risk for oral diseases, such as dental caries and/or mucosal infections.11
The protective qualities of saliva depend on volume. These protective qualities can be significantly enhanced or reduced, depending on the rate of secretion in unstimulated and stimulated conditions.1 Unstimulated, normal salivary secretion is > 0.3 ml/minute, with ranges from 0.5 liter per day to 1.5 liters per day, compared with ranges of 0.1 ml/minute to 0.7 ml/minute in patients with reduced salivary production.3 Decreased salivary flow creates an oral environment that inadequately neutralizes acids, increasing the intraoral pH for long periods.11 Any decrease in salivary volume should be monitored, as it can significantly shift the balance in caries risk. Additionally, other risk factors — such as frequent consumption of acidic drinks and/or high-sugar diet — can accelerate demineralization in an already compromised environment.
ASSESSING SALIVARY FLOW
Assessment of salivary gland function should be included in routine dental visits, as it is essential in diagnosing salivary gland hypofunction as the cause of xerostomia.3,15,16 Clinicians generally use a subjective approach to identify and assess dry mouth, such as a patient’s response to a health questionnaire or medical history form.3 However, an objective measurement of qualitative or quantitative changes in saliva is ideal and best captured by collecting whole saliva or saliva from individual glands.17
Saliva is produced and secreted from the major and minor salivary glands. The major salivary glands are the parotid, submandibular and sublingual. Individual major salivary gland saliva samples can be captured with a modified Carlson-Crittenden device for the parotid gland, and a modified Wolff saliva collector for the submandibular and sublingual glands.3 Whole saliva can be collected in either an unstimulated or stimulated method. Both are collected with a tube and funnel.3 Methods used to stimulate whole saliva flow include gum base, paraffin wax, rubber bands and citric acid.3 The ability to assess salivary flow rate chairside enables clinicians to identify patients experiencing salivary gland hypofunction and develop personalized treatment options to support the remineralization process.
ANALYZING SALIVA SAMPLES
Once saliva samples have been collected, chairside and laboratory tests are available for analysis. These tools can be used to evaluate salivary pH, as well as saliva buffering capacity.18 Research by Singh et al18 evaluated the use of the Saliva-Check Buffer kit, Systronics electrode pH meter, Saliva-Check Mutans kit, and a semi-autoanalyser in identifying caries risk in children. Compared to subjects with active caries lesions, the results indicated the mean values for pH level, buffering capacity, and calcium and phosphorous ions were higher in children without caries.18 Anand et al19 measured pH values and buffering capacity of saliva using a handheld pH meter after hydrochloric acid titration. The results revealed a significant relationship between pH levels and dental caries experience. These studies show chairside and/or laboratory saliva testing can be included in routine dental examinations as a noninvasive method to help predict caries risk.18,19
TREATMENT OPTIONS
Saliva substitutes and stimulants used for salivary gland hypofunction are available in sprays, gels, oils, mouthrinses and chewing gums, and each option differs with respect to the base substance, chemical composition and viscosity — not to mention patient preference.11 Other treatment options directly impact the remineralization process. There is conflicting research as to whether one therapy is better than another.20 Oral health professionals should be knowledgeable about the differences in these options so a customized treatment plan can be created to reduce caries risk and promote remineralization.
Certain salivary substitutes have reportedly increased demineralization by significantly decreasing intraoral pH.21–23 Aykut-Yetkiner et al11 found that four saliva substitutes increased demineralization due to low pH or the presence of citric acid found in the substitute. However, several saliva substitutes were found to significantly increase remineralization due to the high-viscous consistency of the products, leading to mechanical protection of the tooth surface.11 Patients with xerostomia may wish to use high-viscous saliva substitutes and avoid saliva substitutes with low pH or citric acid.11
In combination with at-home fluoride products, the use of professionally applied fluoride may enhance remineralization. Poor salivary contact and limited accessibility to interproximal surfaces make it difficult to manage incipient caries lesions.24 Songsiripradubboon et al24 investigated the remineralization properties of fluoride mouthrinses (used at different times in conjunction with fluoride toothpaste) on incipient lesions. Results showed that twice-daily use of 0.05% sodium fluoride mouthrinse, combined with twice-daily use of fluoride toothpaste, facilitated the greatest remineralization of incipient caries.
The remineralization process in patients with reduced salivary production is often hindered, and the use of fluoride can be limited by the lack of calcium and phosphate ions present.25 Fluoride, calcium and phosphate are needed to aid in the remineralization process during a cariogenic attack.26 Amorphous calcium phosphate (ACP), a combination of soluble salts of calcium and phosphorous, may help to remineralize tooth structure.27 Peric et al28 evaluated the effect of casein phosphopeptide-ACP (CPP-ACP) and casein phosphopeptide-amorphous calcium fluoride phosphate (CPP-ACFP) pastes in patients with Sjögren’s syndrome. The results indicated that patients who used these pastes experienced a slight increase in salivary pH values, a significant rise in plaque pH values, and partial or complete occlusion of enamel defects. Evidence suggests that pastes containing CPP-ACP/CPP-ACFP enhance remineralization in patients with Sjögren’s syndrome.26 Mendes et al29 evaluated the effect of paste containing CPP-ACP and paste containing CPP-ACP combined with fluoride in the remineralization of white-spot lesions. The results showed the highest rates of remineralization occurred with the use of CPP-ACP combined fluoride paste. This clinical trial suggests CPP-ACP pastes containing fluoride may increase the efficacy of CPP-ACP in the remineralization process.29
Other options used to support remineralization include calcium sodium phosphosilicate and tri-calcium phosphate. Calcium sodium phosphosilicate is composed of calcium, sodium, phosphorus and silica, and is designed to release calcium and phosphate, enhancing remineralization.30 Used in collaboration with fluoride, tri-calcium phosphate may support remineralization better than fluoride alone.31
TRANSLATION INTO CLINICAL PRACTICE
Saliva’s role in the demineralization/remineralization process significantly impacts caries management and risk assessment protocols. It is imperative that all members of the dental team understand the progression of dental caries is a dynamic process alternating between demineralization and remineralization, and work to implement appropriate assessment and management strategies.32 In a normal physiological environment (absent from salivary gland dysfunction), saliva has the ability to neutralize the acid produced by plaque biofilm.33 Once combined with fluoride, the calcium and phosphate ions found in saliva will have a good probability of arresting caries during the remineralization process.33
As noted, the caries process results from the interaction of protective and pathologic factors in saliva and plaque biofilm, as well as the amounts of cariogenic and noncariogenic bacteria present.33 The ease of access to saliva provides a noninvasive method for monitoring the progression of caries lesions, and supports a personalized approach to caries management and risk assessment.32,34 In turn, this can prompt efforts to remineralize noncavitated lesions, thus minimizing the need for restorative treatment.32 Previous research has shown that restoring teeth alone does not arrest dental caries or prevent future lesions.35 Clinicians must assess and identify when a patient does not have a normal physiological oral environment, whether from reduced salivary gland function or behavioral risks. This can be done via a variety of chairside tests that assess levels of Steptococcus mutans, pH, buffering capacity, salivary flow, and the adenosine triphosphate levels of the patient’s biofilm.34
An evidenced-based approach to caries management and prevention involves altering the behavior complex of plaque biofilm and transforming risk factors to promote oral health.36–38 This approach favors risk assessment and prevention rather than surgical/restorative treatment.39–41 The ICDAS assessment tool was developed as a means to assist clinicians, researchers and epidemiologists in consistently detecting caries by determining the approximate depth and extent of lesions via clinical visualization.42 Additionally, ICDAS was developed with the intent to reduce unnecessary surgical/restorative treatment. The system notes that even demineralization that has penetrated the dentin with intact enamel can be remineralized. Such lesions have a visible shadow under the noncavitated enamel surface in both pits and fissures, as well as smooth surfaces.42
Typically, several issues must be addressed at once to successfully transform an individual’s risk factors — such as oral bacterial levels, absence of saliva, and lifestyle.33 Caries risk assessment tools, including the Caries Management by Risk Assessment protocol, help clinicians collect information about individual risk and protective factors that can help identify future caries risk.33 Combining caries risk assessment, chairside salivary diagnostics, and regular hard tissue examinations using validated systems (including ICDAS) allows clinicians to prevent lesions or identify noncavitated lesions in their earliest stages. This also supports the implementation of individualized plans for remineralization and caries management, and should include consideration of sialagogues for comfort, in addition to xylitol, prescription fluorides, additional chairside fluoride treatments, and other adjunctive means to promote remineralization.33,43
CONCLUSION
Saliva is an important biological and protective factor in the remineralization process.1 The pH level of saliva, along with the amount of available calcium and phosphate, directly impact demineralization during acidic challenges.4 Early caries lesions can be detected and reversed with the help of noninvasive chairside tools to assess salivary production, pH levels and buffering capacity. Additionally, saliva substitutes can help support remineralization in patients experiencing salivary gland hypofunction.11 Oral health professionals are advised to be knowledgeable about the various preventive therapies available to enhance remineralization, and use this information to create personalized treatment plans to reduce dental caries risk and improve patients’ oral health.
REFERENCES
- Hara AT, Zero DT. The potential of saliva in protecting against dental erosion. Monogr Oral Sci. 2014;25:197–205.
- Meurman JH, ten Cate J. Pathogenesis and modifying factors of dental erosion. Eur J Oral Sci. 1996;104:199–206.
- Navazesh M, Kumar SK, University of Southern California School of Dentistry. Measuring salivary flow: challenges and opportunities. J Am Dent Assoc. 2008;139:35S–40S.
- Aiuchi H, Kitasako Y, Fukuda Y, et al. Relationship between quantitative assessments of salivary buffering capacity and ion activity product for hydroxyapatite in relation to cariogenic potential. Aust Dent J. 2008;53:167–171.
- Abou Neel EA, Aljabo A, Strange A, et al. Demineralization-remineralization dynamics in teeth and bone. Int J Nanomedicine. 2016;11:4743–4763.
- Creeth JE, Karwal R, Hara AT, Zero DT. A randomized in situ clinical study of fluoride dentifrices on enamel remineralization and resistance to demineralization: effects of zinc. Caries Res. 2018;52:129–138.
- Featherstone JD. Dental caries: a dynamic disease process. Aust Dent J. 2008;53:286–291.
- Pretty IA, Ellwood RP. The caries continuum: opportunities to detect, treat, and monitor the remineralization of early caries lesions. J Dent. 2013;41:S12–S21.
- Aranibar Quiroz EM, Alstad T, Campus G, Birkhed D, Lingstrom P. Relationship between plaque pH and different caries-associated variables in a group of adolescents with varying caries prevalence. Caries Res. 2014;48:147–153.
- Shivakumar KM, Prasad S, Chandu GN. International caries detection and assessment system: a new paradigm in detection of dental caries. J Conserv Dent. 2009;12:10–16.
- Aykut-Yetkiner A, Wiegand A, Attin T. The effect of saliva substitutes on enamel erosion in vitro. J Dent. 2014;42:720–725.
- Flink H, Bergdahl M, Tegelberg A, Rosenblad A, Lagerlöf F. Prevalence of hyposalivation in relation to general health, body mass index and remaining teeth in different age groups of adults. Community Dent Oral Epidemiol. 2008;36:523–531.
- Delli K, Spijkervet FK, Kroese FG, Bootsma H, Vissink A. Xerostomia. Monogr Oral Sci. 2014;24:109–125.
- Smith RG, Burtner AP: Oral side-effects of the most frequently prescribed drugs. Spec Care Dentist. 1994;14:96–102.
- Ship JA, Fox PC, Baum BJ. How much saliva is enough? “Normal” function defined. J Am Dent Assoc. 1991;122:63–69.
- Navazesh M. Methods for collecting saliva. Acad N Y Acad Sci. 1993;694:72–77.
- Navazesh M, Christensen CM. A comparison of whole mouth resting and stimulated salivary measurement procedures. J Dent Res. 1982;61:1158–1162.
- Singh S, Sharma A, Sood PB, Sood A, Zaidi I, Sinha A. Saliva as a prediction tool for dental caries: an in vivo study. J Oral Biol Craniofac Res. 2015;5:59–64.
- Anand S, Masih U, Yeluri R. Comparative quantitative assessments of salivary ion activity product for hydroxyapatite and buffering capacity in children with different caries experience. J Clin Pediatr Dent. 2016;40:480–485.
- Furness S, Glenny AM, Worthington HV, et al. Interventions for the treatment of oral cavity and oropharyngeal cancer: chemotherapy. Cochrane Database Syst Rev. 2010;8:CD006386.
- da Silva Marques DN, da Mata AD, Patto JM, et al. Effects of gustatory stimulants of salivary secretion on salivary pH and flow in patients with Sjögren’s syndrome: a randomized controlled trial. J Oral Pathol Med. 2011;40:785–792.
- Jensdottir T, Buchwald C, Nauntofte B, Hansen HS, Bardow A. Erosive potential of calcium-modified acidic candies in irradiated dry mouth patients. Oral Health Prev Dent. 2010;8:173–178.
- Jensdottir T, Nauntofte B, Buchwald C, Bardow A. Effects of sucking acidic candy on whole-mouth saliva composition. Caries Res. 2005;39:468–474.
- Songsiripradubboon S, Hamba H, Trairatvorakul C, Tagami J. Sodium fluoride mouthrinse used twice daily increased incipient caries lesion remineralization in an in situ model. J Dent. 2014;42:271–278.
- Reynolds EC. Casein phosphopeptide-amorphous calcium phosphate: the scientific evidence. Adv Dent Res. 2009;21:25–29.
- Oliveira PR, Coutinho TC, Portela MB, Paula VC, Tostes MAl. Influence of biofilm formation on the mechanical properties of enamel after treatment with CPP-ACP crème. Braz Oral Res. 2017;31:e84.
- Dorozhkin SV. Amorphous calcium (ortho) phosphates. Acta Biomater. 2010;6:4457–4475.
- Peric T, Markovic D, Petrovic B, et al. Efficacy of pastes containing CPP-ACP and CPP-ACFP in patients with Sjögren’s syndrome. Clin Oral Investig. 2015;19:2153–2165.
- Mendes AC, Restrepo M, Bussaneli D, Zuanon AC. Use of casein amorphous calcium phosphate (CPP-ACP) on white-spot lesions: randomised clinical trial. Oral Health Prev Dent. 2018;16:27–31.
- Burwell AK, Litkowski LJ, Greenspan DC. Calcium sodium phosphosilicate (NovaMin): remineralization potential. Adv Dent Res. 2009;21:35–39.
- Karlinsey RL, Mackey AC, Walker ER, Frederick KE. Preparation, characterization, and in vitro efficacy of an acid-modified β-TCP material for dental hard-tissue remineralization. Acta Biomater. 2010;6:969–978.
- Fontana M, Wolff M. Translating the caries management paradigm into practice: challenges and opportunities. J Calif Dent Assoc. 2011;39:702–708.
- Hurlbutt M, Young DA. A best practices approach to caries management. J Evid Based Dent Pract. 2014;14(Suppl):77-86.
- Nový BB. Saliva and biofilm-based diagnostics: a critical review of the literature concerning sialochemistry. J Evid Based Dent Pract. 2014;14(Suppl):27–32.
- Featherstone JD, White JM, Hoover CI, et al. A randomized clinical trial of anticaries therapies targeted according to risk assessment (caries management by risk assessment). Caries Res. 2012;46:118–129.
- Marsh PD. Microbiology of dental plaque biofilms and their role in oral health and caries. Dent Clin North Am. 2010;54:441–454.
- Young DA, Featherstone JD, Roth JR. Curing the silent epidemic: caries management in the 21st century and beyond. J Calif Dent Assoc. 2007;35:681–685.
- Hara AT, Zero DT. The caries environment: saliva, pellicle, diet, and hard tissue ultrastructure. Dent Clin North Am. 2010;54:455–467.
- Yorty JS, Walls AT, Wearden S. Caries risk assessment/ treatment programs in U.S. dental schools: an eleven-year follow-up. J Dent Educ. 2011;75:62–67.
- Maupome G, Isyutina O. Dental students’ and faculty members’ concepts and emotions associated with a caries risk assessment program. J Dent Educ. 2013;77:1477–1487.
- Teich ST, Demko C, Al-rawi W, Gutberg T. Assessment of implementation of a CAMBRA-based program in a dental school environment. J Dent Educ. 2013;77:438–447.
- Dikmen, B. ICDAS II criteria (international caries detection and assessment system). J Istanb Univ Fac Dent. 2015;49:63–72.
- Nassar HM, Lippert F, Eckert GJ, Hara AT. Dentifrice fluoride and abrasivity interplay on artificial caries lesions. Caries Res. 2014;48:557–565.
Featured image by PHOTON-PHOTOS/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. April 2019;5(4):38–41.