Association Between Human Papillomavirus and Oral Health Reading Materials
This course was published in the July 2017 issue and expires July 2020. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Identify the prevalence and incidence of oral human papillomavirus (HPV) in the United States.
- Discuss the safety and effectiveness of the HPV vaccine.
- Explain the role of oral health professionals in discussing the HPV vaccine in dental settings.
The human papillomavirus (HPV) is one of the most common sexually transmitted infections. It is so common, in fact, that nearly all sexually active men and women will be infected at some point in their lives.1 HPV of the oral cavity can occur when the virus enters the body, usually through a cut or small tear inside the squamous cells of the oral cavity lined by mucous membranes, such as the inner aspects of the mouth, throat, tongue or tonsils. Transmission occurs when these areas come into contact with a virus, allowing it to transfer between epithelial cells. Oral HPV is associated with a variety of oral lesions, cancers of the mouth and oropharyngeal cancers (OPC).2 Poor oral health is an independent risk factor for oral HPV infection.2 While it has been established that oral sexual contact can transfer the HPV virus, it is still poorly understood what other pathways exist. The leading cause of OPC is HPV, resulting in approximately 72% of OPCs in men and 63% of OPCs in women.3,4 Overall, oral cavity cancer (OCC) has steadily decreased in the United States since 1973; however, the prevalence of OPC has increased.5 This may be due to the increasing prevalence of HPV.
HPV strains are classified as high risk (those considered necessary for the development of a cancer) or low risk. HPV 16 and 18, among others, are considered high risk and are the two most commonly associated with oral HPV.6 Each day, approximately 12,000 individuals between the ages of 15 and 24 are affected with HPV, and about 23 million have an oral HPV infection on any given day.1 Of these, approximately 230,000 are HPV 16 and 18.1 Infection is about three times more common in men than women.1
The U.S. Food and Drug Administration (FDA) licensed HPV quadrivalent vaccines in 2006 to protect against HPV 6, 11, 16 and 18.7 The vaccine was aimed at preventing cervical cancers and genital warts in women and girls. In 2009, the FDA licensed a bivalent vaccine to protect against HPV 16 and 18 in women and girls, and maintained the quadrivalent vaccine for men to prevent genital warts. The 9-valent (9vHPV) vaccine was approved for both males and females in 2014.8 The 9vHPV includes the HPV types in the quadrivalent HPV (qHPV) vaccine (6, 11, 16 and 18) and five additional oncogenic types (31, 33, 45, 52 and 58). This paper will focus on 9vHPV because it has the potential to increase overall cancer prevention from 70% to 90%; in addition, it is approved for use in both boys and girls.8
The Advisory Committee on Immunization Practices (ACIP) recommends that routine HPV vaccination be initiated at age 11 or 12 for girls and boys. Vaccination is also recommended for females age 13 to 26 and males age 13 to 21 who have not been vaccinated previously.9 In 2016, new recommendations in support of a two-dose schedule for girls and boys who initiate the vaccination series between the ages of 9 and 14 were published.10 Three doses are recommended for those who initiate the vaccination series at age 15 to 26 and for immunocompromised individuals.10 Vaccination is also recommended up to age 26 for men who have sex with men and for immunocompromised individuals (including those with human immunodeficiency virus) if not vaccinated previously. The vaccines are most effective when given to children before they become sexually active. They are not recommended for those with a history of severe allergic reactions to any HPV vaccine components or women who are pregnant.7
PREVALENCE AND INCIDENCE
While the incidence of conventional tobacco-associated squamous cell carcinoma has decreased over the past 30 years, the incidence of OCC and other head and neck cancers has increased.11,12 This is attributed to changes in social behaviors — especially in adolescents — leading to increasing numbers of HPV infections.11–14
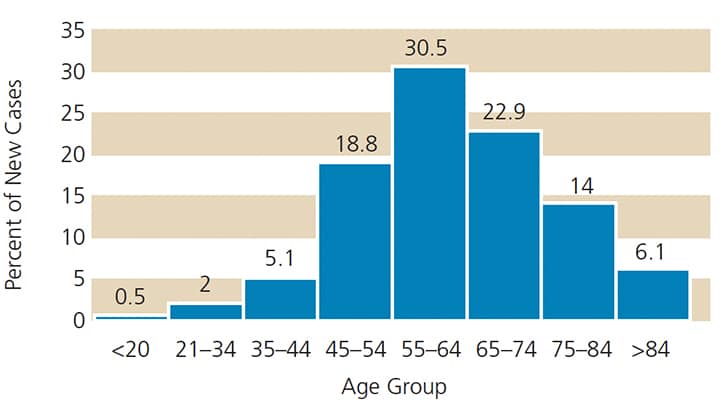
Source: SEER 8 2009–2013, all races, both sexes
HPV affects approximately 80 million Americans,15 and each year nearly 14 million individuals become infected.16 HPV causes 30,700 occurrences of cancer annually in the United States, including cancer of the vagina, vulva, penis, anus, rectum and oropharynx. The HPV vaccination can prevent most of these cancers.16 HPV is the leading cause of OPC and contributes to a smaller number of OCC cases. In 2017, the number of new HPV-related OPC cases is expected to rise to 49,670.17 While the mean age at diagnosis is 62, the number of cases in young adults and adolescents is on the rise (Figure 1). Although the death rate for OCC has remained at approximately 57%, the number of new cases has been rising about 0.6% per year over the past 10 years (Figure 2).15

Source: SEER 9 Incidence 1975–2013, all races, both sexes, rates are age adjusted
HPV-associated cancer is a specific cellular type that is diagnosed in a part of the body where HPV is found. An HPV-attributable cancer is one that is more than likely caused by HPV. A study that used population-based data tested cancer tissue to estimate the percentage of cancers that were attributable to HPV. It concluded that many were HPV-attributed OPCs.16 To determine the number of HPV-attributable cancers, the number of HPV-associated cancers was multiplied by the percentage of cancers probably caused by HPV. Based on this definition, about 70% of all OPCs are attributable to HPV, with 60% being attributed to HPV types 16 and 18, while 6% can be attributed to HPV types 31, 33, 45, 52 and 58.18
EDUCATING PROVIDERS AND PATIENTS
Oral health professionals play an important role in the prevention of oral HPV. Clinicians must attain foundational knowledge of basic clinical medicine to safely and effectively treat individuals with a variety of common systemic diseases. Dental providers have the knowledge and experience to care for patients who present with a variety of conditions. Because many primary care activities are conducted in a dental office — such as screening for diabetes and managing hypertension — dental teams are well positioned to be health care educators.
Early detection of OCCs is key to reducing morbidity. The American Dental Association recommends that oral health professionals conduct secondary preventive activities to screen for oral cancers; however, little evidence supports the efficacy of these efforts.19 That said, significant evidence supports the HPV vaccine as a preventive modality against HPV-associated cancers.
A 2015 national survey found that 63% of girls and 50% of boys age 13 to 17 had begun their HPV vaccine series, with approximately 50% of girls and 28.1% of boys completing the series.18 Figure 3 shows the national coverage of HPV vaccination.20
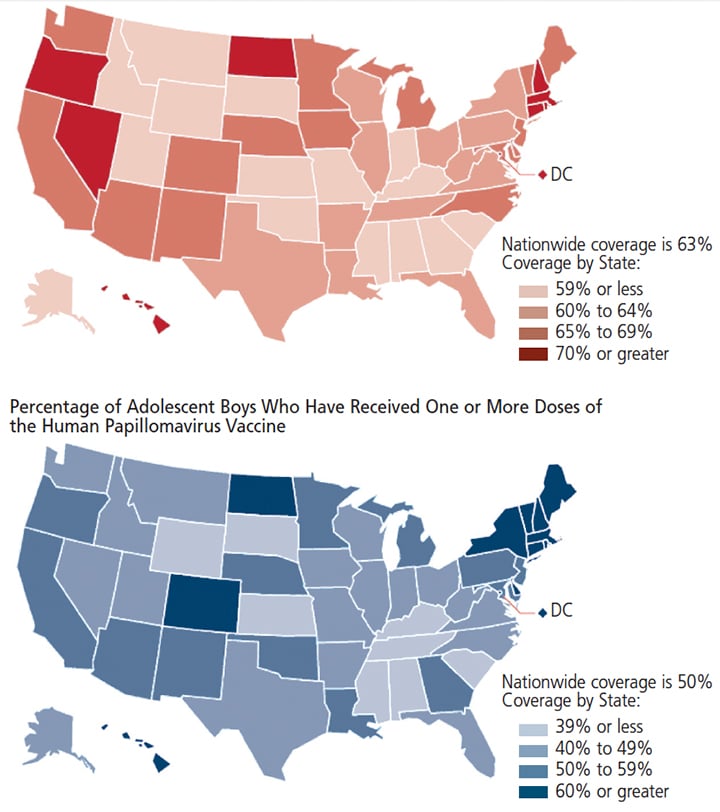
papillomavirus vaccine among adolescents age 13 to 17 in the United States.20
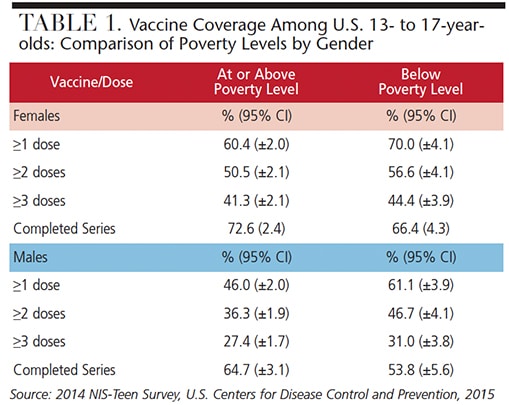
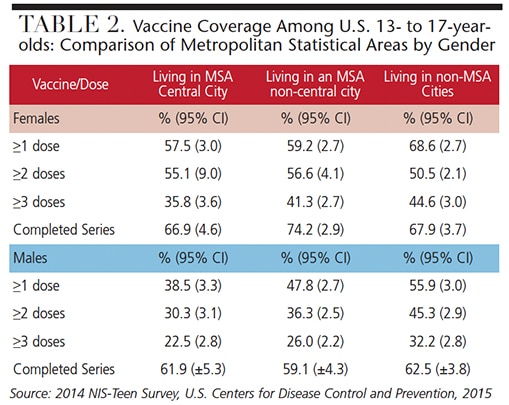
Table 1 and Table 2 provide information about vaccine coverage among individuals age 13 to 17 by gender and poverty level (Table 1), and gender and Metropolitan Statistical Area (Table 2). There could be many reasons why Americans fall short of vaccination goals. Some of the perceived objections include the belief that vaccines contain toxins that can be harmful, that vaccines don’t really provide protection from diseases, vaccines are just a means for doctors and pharmaceutical companies to make money, potential side effects may be worse than the actual disease, and, in the case of HPV, the sensitivity of discussing with patients (or parents and caregivers) how HPV is contracted.
In order to improve compliance with HPV vaccination recommendations, oral health professionals should be prepared to discuss the importance of immunizations with patients. Currently, practice behavior guidelines regarding counseling patients on the benefits of the HPV vaccine are not available. Oral health professionals, however, should become familiar with HPV and its connection to OPC.21 Because of the science confirming the association between OPC and HPV, the roles of oral health professionals will continue to expand in this arena. A recent study found a knowledge deficit among oral health professionals regarding HPV.4 While there was no significant association found between the level of knowledge and amount of communication frequency with patients regarding HPV and OCC, the authors suggested there is a need to improve health literacy regarding HPV and oral cancer among oral health professionals.4
HPV knowledge gaps exist among patients with regard to prevention, treatment, and health consequences, even in those with adequate health literacy.22 Testing methods for oral HPV infection and the potential benefits of the HPV vaccine were also areas in which patients were deficient. The knowledge gap in both dental providers and patients creates a new challenge. The trust patients have in oral health professionals highlights the importance of providing critical health care information in dental settings.
ADOPT A PROACTIVE APPROACH
As an example of a proactive approach to improving HPV literacy, in Nevada, only 24% of males received all doses of the HPV vaccine in 2015, compared with 43% of females — yet these figures represent significant improvement from 2013 (7% for males, 27% for females).23 The increase can be attributed to the interdisciplinary health promotion efforts of the Nevada State Immunization Program, Immunize Nevada (a statewide immunization coalition) and a diverse group of community stakeholders.24 Immunize Nevada began offering continuing education courses and webinars to health care providers with the support of the U.S. Centers for Disease Control and Prevention (CDC) and Nevada Area Health Education Centers.25 Immunize Nevada plays a key role in the planning of the Nevada Health Conference, which includes dedicated time for education on vaccinations and health promotion strategies to increase vaccination rates.
In addition to online training options, health care professionals are encouraged to attend the immunization clinics scheduled at local health fairs, which are excellent opportunities for parents to receive information about — and obtain — the HPV vaccine and oral health screenings for children. Establishing collaborative partnerships with other professionals — including immunization staff — can help raise public awareness about signs, symptoms, risk factors, and changes in the demographics of OCC and OPC. Creating an interprofessional workforce that includes medical and dental professionals to assist in counseling patients on the benefits of the HPV vaccine will help reduce the risk of HPV-related oral cancers. Offering oral cancer screenings in targeted, high-risk populations during an immunization clinic or health fair creates an opportunity to discuss the increasing trends of OCC/OPC and HPV.
Integrating effective communication strategies within clinical settings can build patient awareness to the possible risks of HPV-related OCC and OPC. Health care professionals must feel comfortable discussing HPV and the HPV vaccine with patients. While it is difficult to discuss sensitive medical concerns in a health care setting, rephrasing the message as a cancer-prevention strategy can encourage future conversations with patients. Webinars and continuing education courses can assist health care providers in the most effective communication tools. Integrating HPV education into dental and dental hygiene curricula will also help prepare future providers in addressing HPV and the HPV vaccine in dental settings. The next generation of health care professionals needs to be well versed in the emerging evidence as it relates to OCC/OPC and HPV.
VACCINE INFORMATION
Parents and caregivers should be asked whether their adolescent or young adult children have received the HPV vaccine at the time the medical history is completed. Due to the perceived sensitive nature of the infection, it may be difficult to broach this subject in a dental setting, but, as noted, phrasing the topic as a cancer-prevention strategy helps facilitate discussion. Providers can also create an automatic note within the health record to encourage questioning about the initial dose of the HPV vaccine and completion of the recommended doses. Educating patients and providing a resource sheet that outlines additional information on the benefits and risks of the HPV vaccine and its efficacy in the prevention of a variety of cancers is helpful.
Increasing awareness about the HPV vaccine is a positive step, but current data do not support the prevention of OPCs with the vaccine unequivocally. Published data relative to the long-term effects of the HPV vaccine on OCC and OPC are sparse. The published literature, however, confirms this topic has important research and public health implications. The CDC has carefully studied the risks and benefits of HPV vaccination. The vaccine is recommended because the benefits — such as cancer prevention — far outweigh the risks of side effects.26 Nearly 90 million doses of HPV vaccines were distributed in the United States from June 2006 through March 2016, with the most frequently reported symptoms being fainting, dizziness, headache, nausea, fever and pain, redness, and swelling in the arm where the injection was given. The CDC notes that some populations are contraindicated for the vaccine, including:26
- Anyone who has had a life-threatening allergic reaction to any component of HPV vaccine, or to a previous dose of HPV vaccine
- Individuals considering the Gardasil or Gardasil 9 vaccine should inform their health care provider about any severe allergies, including an allergy to yeast
- Individuals considering the Cervarix vaccine should inform their health care provider about any severe allergies, including an allergy to latex
- Pregnant women — although women who are breastfeeding may get the vaccine
- Patients with a moderate or severe illness should wait until they are well; individuals who are mildly ill (low-grade fever of less than 101°, cold, runny nose or cough) can still be vaccinated
SUMMARY
According to the CDC, the HPV vaccine is safe, effective and offers long-lasting protection against HPV-associated cancers. Vaccine recommendations are based on characteristics of the immunobiologic product, existing scientific evidence, epidemiology, current safety concerns, published literature and expert opinion. There are benefits and risks associated with all types of vaccines, and the HPV vaccine is no different. Therefore, experts attempt to balance the science against the potential risks prior to making recommendations. The HPV vaccine is new, especially to dental providers. The relative balance can change, which is why health care professionals should stay abreast of the most current recommendations. More longitudinal studies are needed on the effect of HPV vaccine on OPC. Clinicians are charged with keeping up to date on the research regarding HPV and oral cancers, as well as the effectiveness and safety of the HPV vaccination, so they can appropriately advise patients.
REFERENCES
- U.S. Centers for Disease Control and Prevention. General HPV Infection Fact Sheet. Available at: cdc.gov/std/hpv/stdfact-hpv.htm. Accessed May 17, 2017.
- Bui TC, Markham CM, Ross MW, Mullen PD. Examining the association between oral health and oral HPV infection. Cancer Prev Res (Phila). 2013;6:917–924.
- Viens LJ, Henley SJ, Watson M, et al. Human papillomavirus-associated cancers—United States, 2008-2012. MMWR Morb Mortal Wkly Rep. 2016;65:661–666.
- Daley EM, Thompson EL, Vamos CA, et al. HPV-related knowledge among dentists and dental hygienists. J Cancer Educ. December 30, 2016. Epub ahead of print.
- Denson L, Janitz AE, Brame LS, Campbell JE. Oral cavity and oropharyngeal cancer: Changing trends in incidence in the United States and Oklahoma. J Okla State Med Assoc. 2016:109:339–345.
- U.S. Centers for Disease Control and Prevention. Epidemiology and Prevention of Vaccine-Preventable Diseases. Available at:cdc.gov/vaccines/pubs/pinkbook/hpv.html. Accessed May 17, 2017.
- U.S. Centers for Disease Control and Prevention. FDA licensure of bivalent human papillomavirus vaccine (HPV2, Cervarix) for use in females and updated HPV vaccination recommendations from the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2010;59:626–629.
- Markowitz LE, Dunne EF, Saraiya M, et al. Human papillomavirus vaccination: recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2014;63:1–30.
- Petrosky E, Bocchini JA, Hariri S, et al. Use of 9-valent human papillomavirus (hpv) vaccine: updated HPV vaccination recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep.2015;64:300–304.
- Meites E, Kempe A, Markowitz LE. Use of a 2-dose schedule for human papillomavirus vaccination—updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2016;65:1405–1408.
- Denson L, Janitz AE, Brame LS, Campbell JE. Oral cavity and oropharyngeal cancer: Changing trends in incidence in the US and Oklahoma. J Okla State MedAssoc. 2016;109:339–344.
- Mehta V, Yu GP, Schantz SP. Population-based analysis of oral and oropharyngeal carcinoma: changing trends of histopathologic differentiation, survival, and patient demographics. Laryngoscope. 2010;120:2003–2012.
- Reagan-Steiner S, Yankey D, Jeyarajah J, et al. National, regional, state, and selected local area vaccination coverage among adolescents aged 13–17 years—United States, 2015. MMWR Morb Mortal Wkly Rep. 2016;65:850–858.
- Gayar OH, Rutervusch JJ, Elshaikh M, et al. Oropharyngeal carcinoma in young adults: an alarming national trend. Otolaryngol Head Neck Surg. 2014:150:594–601.
- Oral Cancer Foundation. The HPV connection. Available at: oral cancer foundation.org/ understanding/hpv/. Accessed May 17, 2017.
- Saraiya M, Unger ER, Thompson TD, et al. US assessment of HPV types in cancers: implications for current and 9-valent HPV vaccines. J Natl Cancer Inst.2015;107:djv086.
- National Cancer Institute. Surveillance, Epidemiology, and End Results Program.Available at: seer.cancer.gov/seerstat. Accessed May 17, 2017.
- U.S. Centers for Disease Control and Prevention. HPV and Cancer. Available at: cdc.gov/cancer/hpv/statistics/cases.htm. Accessed May 17, 2017.
- Rethman MP, Carpenter W, Cohen EEW, et al. Evidence-based clinical recommendations regarding screening for oral squamous cell carcinomas. J Am Dent Assoc. 2010;141:509–520.
- U.S. Centers for Disease Control and Prevention. HPV Vaccine Coverage Maps — Infographic. Available at: cdc.gov/hpv/infographics/vacc-coverage.html. Accessed May 17, 2017.
- Hayes C. HPV-changing the face of head and neck cancer and dental societies’ responsibilities. Today’s FDA. 2015:27:38–39.
- Inglehart RC, Taberna M, Pickard PKL, et al. HPV knowledge gaps and information seeking by oral cancer patients. Oral Oncol. 2016;63:23–29.
- Nevada Division of Public and Behavioral Health/Nevada Immunization Program. Available at: dpbh.nv.gov/Programs/Immunizations. Accessed May 17, 2017.
- Immunize Nevada. Community Impact. Available at: immunizenevada.org. Accessed May 17, 2017.
- Nevada Area Health Education Centers. Statewide Initiatives. Available at: med.unr.edu/statewide/ahec. Accessed May 17, 2017.
- U.S. Centers for Disease Control and Prevention. HPV Vaccine Safety. Available at: cdc.gov/vaccinesafety/vaccines/hpv/hpv-safety-faqs.html#A10. 2017. Accessed May 17, 2017.
FEATURED IMAGE BY LUCKYRACCOON/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. July 2017;3(7):32–36