Evaluating Dental Asepsis Programs
Ongoing assessment of office protocols will reinforce optimal infection control practices and help identify areas that need improvement.
Promoting a safe dental visit includes the implementation and regular review of a high-quality infection prevention program to protect patients and staff. An annual evaluation of asepsis efforts can include documentation of standard operating procedures, along with direct observation of infection control protocols used by the staff in the delivery of care.1 Any deficiencies or concerns identified during an evaluation present an opportunity to improve the overall infection control program in a dental facility. Clinicians can implement improvements and promote procedures that are useful, practical and relevant in the delivery of dental care. This review will emphasize effective and attainable program evaluation strategies that include record keeping and evaluation via direct observation.
Ineffective record keeping is a shortcoming of infection control evaluation that detracts from an otherwise effective program. For example, DePaoloa et al2 evaluated compliance with the federal Occupational Safety and Health Association (OSHA) Bloodborne Pathogens Standard and Hazard Communication Program by direct observation of more than 100 dental offices in Maryland. The team reported that “most dental offices inspected complied with the infection control safety issues of greatest concern to staff and patients. These issues included handwashing, use of appropriate personal protective equipment, barrier techniques, and heat sterilization of handpieces, instruments and devices used in dental treatment.” Yet, even though staff adhered to effective practices, record keeping was not optimal, as the authors noted “a greater proportion of dental offices did not comply with [OSHA’s] Bloodborne Pathogens Standard and Hazard Communication Program documentation and record keeping requirements.”2
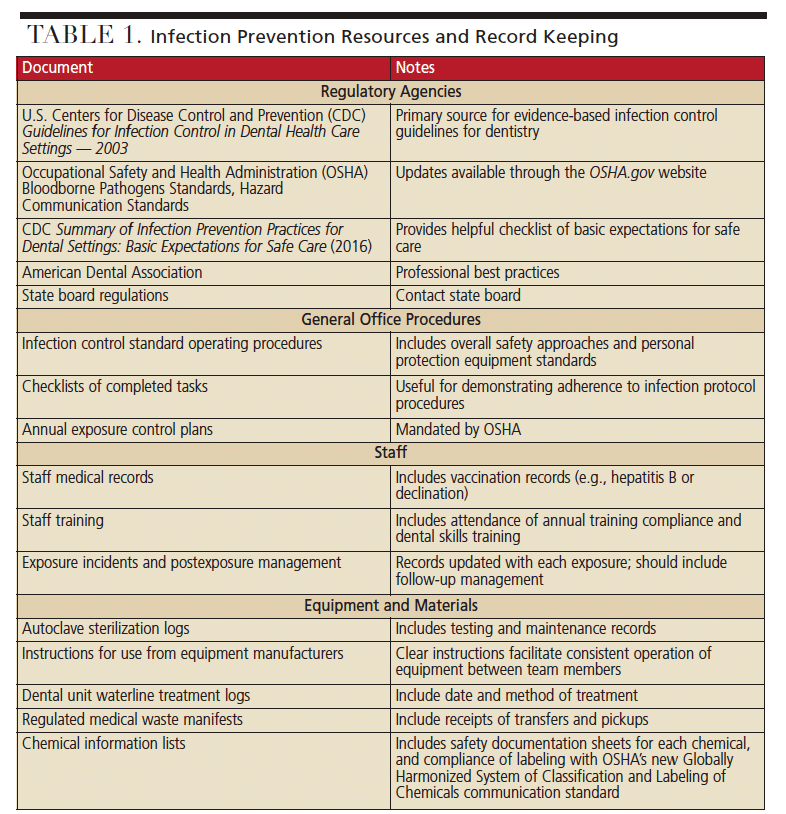
Infection control documentation can be requested by regulatory agencies, and inadequate record keeping raises questions regarding the effectiveness of an asepsis program. Standards and best practices of documentation can vary based on federal and state laws, but commonly include records regarding office procedures, staff members and equipment (examples are included in Table 1). For example, records of office protocols can include standard operating procedures for infection control, along with checklists of completed tasks (e.g., sterile processing of handpieces). Additionally, office procedures must include a postexposure control plan that is updated annually, with exposure incidents and postexposure management.3
Records of staff members can include adequate and updated staff medical records and vaccination records, including hepatitis B vaccination or declination, and postvaccination titer testing. Additional documentation includes records of compliance with training plans. Records of equipment can include sterilization and testing logs for each autoclave, along with instructions for use, as well as dental unit waterline treatment and testing logs. Materials should also be documented, including regulated medical waste manifests and transfer pickup receipts, chemical information lists of chemicals used, along with their appropriate safety data sheets and compliance with labeling under the new Globally Harmonized System of Classification and Labeling of Chemicals.4
The U.S. Centers for Disease Control and Prevention (CDC) and OSHA produce regulatory documents that should be included in an office’s infection control plan. Printed copies or clear access to digital copies help demonstrate that staff is aware of federal regulatory standards as a basis for compliance. Examples include the CDC Guidelines for Infection Control in Dental Health Care Settings — 2003,5 OSHA Bloodborne Pathogens Standards, Hazard Communication Standards,3 and the CDC Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care.6 Failure to produce such documentation from federal or national agencies can indicate a lack of knowledge of regulations. It can also raise questions about whether staff members are following effective work practices to keep patients and employees safe.
EVALUATION AND COORDINATION
Following effective documentation, the question remains: Is there routine evaluation of an infection control program, including evaluation of performance indicators? A helpful approach is to designate a staff member as infection control coordinator. The coordinator can assess the program through direct observation and evaluate the effectiveness of the office’s asepsis protocol. It is necessary for coordinators and the rest of the dental team to work together to assess and evaluate effectiveness, compliance and opportunities for improvement.
A simple approach for direct observation and assessment is the Infection Prevention Checklist of Dental Settings: Basic Expectations for Safe Care, available from the CDC website (cdc.org/oralhealth).6 The checklist can be used to evaluate the effectiveness of an infection control plan and help ensure the dental health care setting has appropriate infection prevention policies and practices in place. Such practices include appropriate training and education of dental health care personnel in infection prevention practices, and adequate supplies to allow safe care and a safe working environment. The checklist, along with direct observation, can help systematically assess compliance with the expected infection prevention practices and be a foundation for feedback regarding performance. Assessment of compliance should be conducted by direct observation of personnel during the performance of duties and never by the interview process alone.
A key aspect of evaluation is to identify any issues that need attention. To ensure everyone is involved in the process, a team huddle or other staff meeting can be used to go over the evaluation, identify the issues, and set priorities for program improvement.7 Memberships in national organizations may help such efforts; for example, the Organization for Safety, Asepsis and Prevention (OSAP) has a 12-Month Planning Guide to Establish a High-Quality Infection Control and Safety Program, which can be downloaded by members (osap.org).
TEAMWORK COUNTS
While knowing what needs to be done is part of infection prevention, individual actions and team actions speak louder than words. Dental health care personnel must help each other adhere to high-quality infection control procedures and guarantee everyone is using best practices at all times.
To develop an acceptable, relevant and sound program for infection prevention in dental facilities, clinicians can look to the Framework for Program Evaluation in Public Health published by the CDC.8 These recommendations provide standards for effective program evaluation of dental infection control. According to the CDC, “although informal evaluation occurs through routine practice, standards exist to assess whether a set of evaluative activities are well-designed and working to their potential. The Joint Committee on Standards for Educational Evaluation has developed program evaluation standards for this purpose. These standards are designed to assess evaluation of educational programs, but are also relevant for public health programs.” While focused on in-patient settings, the joint committee report includes helpful evaluation standards that can be applied to private practice.9
Standards are grouped into four categories — utility, feasibility, propriety and accuracy — that are used together throughout the evaluation process.
Standard 1: Utility includes the state of being useful or profitable, particularly through being able to perform several functions. The joint commission states, “Utility standards ensure that the evaluation is viable and pragmatic. The use of resources in conducting the evaluation should be prudent and produce valuable findings.”
Standard 2: Feasibility of evaluation includes practices that are viable, pragmatic, and employed as practical and nondisruptive procedures. Resources used for the evaluation should be prudent and value-oriented.
Standard 3: Propriety refers to conforming to accepted standards of behaviors or morals, the details or rules of behavior conventionally considered to be correct, and the condition of being right, appropriate or fitting. According to the commission, propriety exists if the “evaluation is ethical and conducted with regard for the rights and interests of those involved and affected.” Evaluation of an infection prevention program helps emphasize patient safety and disclosure to affected people. Contacting regulatory agencies may be useful for guidance on disclosing the findings of program evaluations.
Standard 4: Accuracy refers to the quality or state of being correct or precise, and, specifically, the degree to which the result of a measurement, calculation or specification conforms to the standard. Qualitative and quantitative evaluations that are systemically gathered and analyzed can help produce reliable conclusions concerning infection prevention programs.
These four standards help users and evaluators select evaluation strategies that are useful, feasible, ethical and accurate. Evaluation requires a great deal of practice, which builds skills in both the science and art of program evaluation. It is more important for evaluators to strive for developing an optimal strategy rather than waiting for an ideal one. It must be emphasized that infection control coordinators can no longer question whether to evaluate a program. Instead, the appropriate questions are:
- What is the best way to evaluate?
- What is being learned from the evaluation?
- How will lessons learned from evaluations be used to make effective and accountable improvements toward a quality dental infection prevention program and increase compliance with protocols?
EVOLVING WORKPLACE REQUIREMENTS
Clinical practice is changing dramatically as a result of new technologies, and infection prevention is also affected. New dental infection control products are common in the marketplace, and changing techniques, precautions and associated work practices can affect established infection prevention programs — for example, via updated instructions or the use of new equipment or products.
Turnover of oral health care personnel requires ongoing infection control training of new team members. A challenge associated with turnover is that infection prevention procedures may vary between facilities. Asepsis protocols learned in one office may not apply to procedures followed by another practice. There can be a range of experience and knowledge regarding appropriate infection prevention procedures among office personnel. Transitioning oral health care professionals can bring a bias or procedure methodology that may need to be modified to align with accepted practices of infection prevention. Here, too, effective evaluation can help identify points of improvement for existing and new team members.
SUMMARY
Program evaluation is the mechanism that coordinators and clinicians must undertake at least annually to assess the office’s infection prevention protocol.10 Evaluation pertains to policies and documentation, along with actual work performance and practices followed by office staff. It includes checks and balances with existing practices, and must also address new procedures and relevant factors related to standards of care designed to prevent disease transmission in the dental workplace.11
The accepted infection prevention protocols for dental settings are effective in promoting safety, and include the use of personal protective equipment.12 Isolated breaches regarding best practices in asepsis can have profound effects on the safety of patients and dental teams. The adage of first “do no harm” is highly appropriate in this regard, and necessitates compliance with accepted standards and procedures for infection prevention. Appropriate evaluation of asepsis programs provides the opportunity to reinforce optimal practices and identify areas or policies that may need to be improved. For oral health care providers, it is necessary to both know what to do, and do what is known.
REFERENCES
- Organization for Safety, Asepsis and Prevention. From Policy to Practice: OSAP’s Guide to the CDC Guidelines. Annapolis, MD: OSAP; 2016:129–132.
- DePaoloa LG, Ward MA, Grace EG, Goodman HS, Cohen LA. Dental office compliance evaluation for the OSHA Bloodborne Pathogens Standard and Hazard Communication Program. J Am Dent Assoc. 1993;124:61–65.
- U.S. Occupational Safety and Health Administration. Bloodborne Pathogens Standard 29 CFR. 1910.1030. Available at: www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=10051. Accessed May 8, 2018.
- United Nations. A Guide to The Globally Harmonized System of Classification and Labeling of Chemicals (GHS). Available at: www.osha.gov/dsg/hazcom/ghsguideoct05.pdf. Accessed May 8, 2018.
- U.S. Centers for Disease Control and Prevention. MWR Recommendations and Reports. Available at: www.cdc.gov/mmwr/preview/mmwrhtml/rr5217a1.htm. Accessed May 8, 2018
- U.S. Centers for Disease Control and Prevention. Infection Prevention Checklist for Dental Settings, Basic Expectations for Safe Care. Available at: www.cdc.gov/oralhealth/infectioncontrol/pdf/safe-care-checklist-a.pdf. Accessed May 8, 2018.
- Miller CH. OSAP Team Huddle: The plan for establishing a high quality infection control program — building a framework for the safest dental visit. Infection Control In Practice. 2016;15(3):1–8.
- U.S. Centers for Disease Control and Prevention. Framework for program evaluation in public health. MMWR Recomm Rep. 1999;48(RR11):1–40.
- Yarbrough DB, Shula LM, Hopson RK, Caruthers, FA. The Program Evaluation Standards: A Guide for Evaluators and Evaluation Users. 3rd ed. Thousand Oaks, Calif: Corwin Press; 2010.
- Miller CH. OSAP. Team Huddle: The plan for establishing a high quality infection control program – assess your current infection control program. Infection Control In Practice. 2016;15(2):4.
- Molinari JA, Terezhalmy GT. Evaluation of a Practical Dental Infection Control Program. In: Molinari JA, Harte JA, eds. Cottone’s Practical Infection Control in Dentistry. 3rd ed. Baltimore, Md: Lippincott Willians & Wilkins; 2010:271–282.
- Garland KV. Brush up on the basics of PPE. Dimensions of Dental Hygiene. 2017;15(10):23–26.
Featured image by WAVEBREAKMEDIA/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. June 2018;4(6):13–14,17.