
Chemotherapeutics in the Treatment of Periodontal Disease
The use of these adjunctive therapies can enhance the outcome of traditional management strategies.
This course was published in the April 2018 issue and expires April 2021. The authors have no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
- Discuss the challenges that clinicians face in managing periodontal disease.
- List the active ingredients, desirable properties, and mechanisms of action of common periodontal chemotherapeutics.
- Explain the types and role of locally applied antimicrobials/antibiotics in treating chronic periodontitis.
Dental professionals continually seek ways to address oral biofilm and the excessive host response responsible for periodontal disease. While mechanical therapy (including scaling, root planing and power instrumentation) is the gold standard for biofilm disruption and removal, research demonstrates that clinicians rarely, if ever, achieve complete removal of the biofilm pathogens1 that tightly adhere to each other and the oral substrate by means of an extracellular matrix.2,3 Bacteria in biofilms are much more difficult to manage with antimicrobial agents than those dispersed as single cells of the same species. Knowing this helps clinicians understand why antibiotics utilized to address the biofilm, either locally applied or systemically administered, are adjuncts to mechanical therapy, and not a stand-alone treatment. Depending on the patient’s risk factors, an optimal approach might include mechanical disruption, host modulation, and adjunctive use of antiseptics and/or antibiotics. Ultimately, clinicians must determine which, if any, adjunctive agent(s) should be utilized in periodontal treatment.
Chemotherapeutic agents are used to address the microbial challenge and modulate the host response by targeting elevated levels of pro-inflammatory or destructive mediators. The term antimicrobial refers to agents that kill microbes or affect the growth and multiplication of microorganisms.4 Various chemotherapeutic agents, typically in the form of mouthrinses or dentifrices, can help control and reduce supragingival plaque and associated gingivitis. Other agents are available to help control and treat chronic periodontitis, such as locally applied antimicrobials/antibiotics (LAAs) and systemically administered antimicrobials. Desirable qualities in an antimicrobial agent include:
- Effectiveness against specific pathogens
- Nontoxicity to oral tissues
- Will not cause an overgrowth of other organisms
- Does not contribute to the development of bacterial resistance
- Substantivity (remains on-site long enough to be effective)5
In addition, the only U.S. Food and Drug Administration (FDA)-approved host modulatory agent is a systemically administered subantimicrobial dose of doxycycline (20 mg administered twice daily). This host modulatory therapy has been shown to reduce excessive levels of enzymes and cytokines known to drive the breakdown of connective tissue and bone metabolism changes associated with periodontitis.6
MOUTHRINSES AND DENTIFRICES
Therapeutic rinses designed to reduce plaque and gingivitis contain antiseptic agents. These chemical antimicrobial adjuncts are applied topically or subgingivally to tooth surfaces, the sulcus, mucous membranes, wounds, or intact dermal surfaces to destroy microorganisms and inhibit their reproduction or metabolism. While most antiseptics are bactericidal, some are bacteriostatic.
The most studied mouthrinses are those containing essential oils, chlorhexidine gluconate (CHX), cetylpyridinium chloride (CPC) and stannous fluoride (SnF2). These agents have been evaluated in preclinical and clinical trials for effectiveness and safety.7,8 In addition, systematic reviews and meta-analyses help clinicians use evidence-based decision-making when selecting these products.7,8 All are safe when used as directed. Disadvantages of rinses include taste alterations, staining, burning, and an increase in calculus formation.9,10 Only mouthrinses containing essential oils carry the American Dental Association (ADA) Seal of Acceptance for the control of plaque and gingivitis. Although some CHX rinses formerly earned the ADA Seal for the control of plaque and gingivitis, the ADA does not currently provide the seal for prescription products. Chemotherapeutic dentifrices in the United States primarily contain triclosan or SnF2. Here is a brief description of the active ingredients in common chemotherapeutic mouthrinses and dentifrices for the control of plaque and gingivitis.
Chlorhexidine: The antiseptic CHX is one of the most effective antiplaque/antigingivitis agents available to clinicians. A systematic review by Gunsolley7 concluded there is strong evidence supporting the efficacy of CHX and essential oils rinses. Sold in the United States as a prescription mouthrinse at 0.12% concentration, most CHX products contain 11.6% alcohol, although some are alcohol free. The mechanism of action is related to an alteration of bacterial adsorption, a reduction in pellicle formation, and an alteration of the bacterial cell wall, resulting in lysis. A major advantage is that CHX is a very substantive agent, resulting in reductions in plaque biofilm and gingivitis ranging from 22% to 61% and 18% to 44%, respectively.11 Disadvantages include tooth staining, alteration of taste, and an increase in calcified deposits.
Essential oils: Mouthrinses formulated with essential oils — thymol, menthol and eucalyptol combined with methyl salicylate — have been studied extensively.7,12 The alcohol content in most products ranges from 21.0% to 26.9%, and the mechanism of action is related to alteration of the bacterial cell wall. Studies have reported plaque and gingivitis reductions ranging from 14% to 56% and 14% to 39%, respectively.4,7 Adverse effects include a burning sensation with use of alcohol-containing formulations, which is diminished with newer alcohol-free products.
Cetylpyridinium chloride: Classified as a quaternary ammonium compound, CPC rinses’ mechanisms of action include rupture of cell walls, the promotion of cell lysis, decreases in cell metabolism, and interfering with the ability of bacteria to attach to tooth surfaces. Studies have reported inconsistent results, however, which is likely due to differences in formulations.7 One systematic review reported CPC as offering a small, but significant, benefit in plaque reduction.13 Side effects include staining, increased calculus formation and occasional burning.
Stannous fluoride: The formulation of these products varies from 0.63% SnF2 in rinses to 0.045% in dentifrices. Gunsolley7 reported that while dentifrices containing SnF2 offered a statistically significant, but small, antiplaque effect, they produced a statistically and clinically significant effect on gingivitis.7 Although SnF2 has received mixed reviews due to the agent’s instability, a dentifrice containing a stabilized 0.454% SnF2, plus zinc citrate, is designed to reduce calculus and stain. The side effects of SnF2 include staining and altered taste.
Triclosan: In the United States, the broad-spectrum antibacterial agent triclosan (0.03%) is only available in a dentifrice with the copolymer Gantrez (2.0%), which improves efficacy. With the addition of 0.243% sodium fluoride in a silica base, it has demonstrated efficacy in reducing plaque, gingivitis calculus and dental caries. Systematic reviews have shown clinically significant results in plaque and gingivitis reduction.7,14–16 Approved for use by the FDA, triclosan toothpaste has a proven safety record and continues to be monitored by the FDA for any adverse effects.17–19 In addition, a 2014 study of subjects who had used triclosan toothpaste for five years reported that continuous use did not result in the development of a triclosan-resistant bacterial population in the oral biofilm.20
The foregoing products are easy to use and most can be purchased over the counter. Clinicians should use the available evidence and their professional judgment in recommending the most appropriate mouthrinse or dentifrice. All products described above have varying levels of evidence for biofilm control.
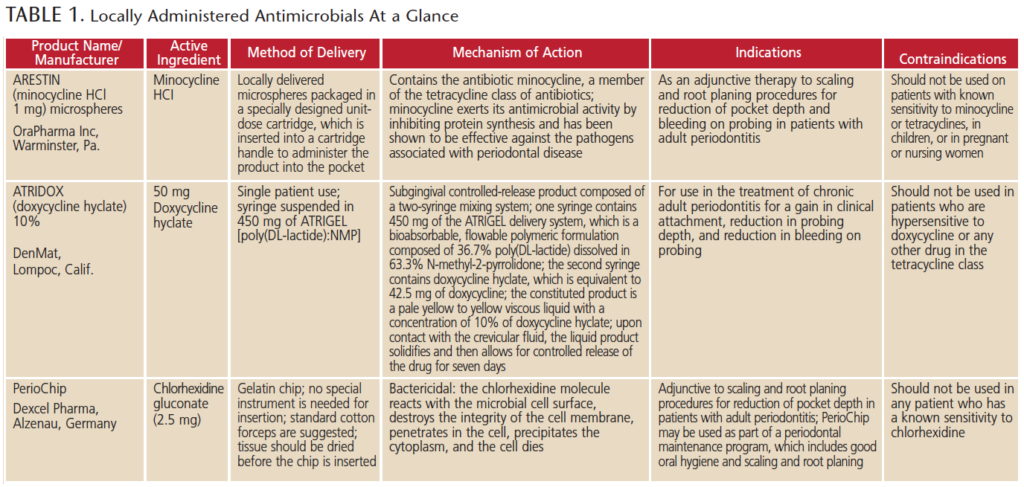
LOCALLY DELIVERED ANTIMICROBIALS
Many patients will respond to thorough debridement and self-care therapies; however, for those who do not improve sufficiently or at all — or continue to decline in periodontal health — stronger adjunctive therapies may be necessary. In these cases, clinicians may consider LAAs. At present, three resorbable, site-specific LAA products are FDA approved for treating chronic periodontitis. These include a chlorhexidine-based chip, minocycline microspheres and a doxycycline hyclate gel (Table 1). The chip product is the only antiseptic LAA; it is not an antibiotic. If clinicians recommend an LAA, patients who have an allergy to the tetracycline class of drugs or who are pregnant should use the chip.
When utilized as an adjunct to scaling and root planing, these agents deliver an antiseptic or antibiotic to the base of the periodontal pocket with the goal of reducing pocket depth and bleeding on probing, while encouraging gains in clinical attachment levels. The substantivity of these agents varies from ~7 days for CHX21 and doxycycline22 to ~14 to 21 days for minocycline,23 and patients benefit from the slow delivery of the active ingredient at a higher minimal inhibitory concentration (MIC) than could be achieved by other applications. Site-specific agents have a higher and longer substantivity, and maintain this MIC long enough to reduce the level of pathogens, leading to improvements in the periodontal condition after single or multiple applications.
While all of these LAAs were studied in clinical trials for the improvement of chronic periodontitis, other investigations report benefits for high-risk patients. For example, Goodson et al24 demonstrated that compared to scaling and root planing alone, minocycline microspheres significantly reduced periodontal pathogens comprising the “red complex” (Porphyromonas gingivalis, Tannerella forsythia and Treponema denticola), particularly in smokers. A 2-year follow-up of doxycycline gel and scaling and root planing in smokers demonstrated sustained improvements in probing depth reductions and attachment level gains beyond what was achieved with scaling and root planing alone.25 Other studies have demonstrated that intensive therapy with minocycline microspheres also results in significant reductions in the overall inflammatory burden, with a reduced risk for cardiovascular events.26,27
To help clinicians with evidence-based decision-making, the ADA Council on Scientific Affairs conducted a systematic review and meta-analysis of nonsurgical treatments of chronic periodontitis utilizing scaling and root planing with or without adjuncts.28 The authors assessed the overall level of evidence as high, moderate or low. A clinical practice guideline was developed that provides recommendations for these treatments based on a scale of strong, in favor, weak, expert opinion for, expert opinion against, and against.29 The chlorhexidine chip was rated weak, and the minocycline microspheres and doxycycline gel received expert opinion for. The authors emphasize that expert opinion for does not imply endorsement, but, rather, signifies that evidence is lacking and the level of certainty of the evidence is low. Clinicians should determine recommendations based on their professional judgment and individual patient needs and preferences.
OTHER LOCALLY APPLIED PRODUCTS
Based on the herbs elder, centella asiatica and purple coneflower, the novel PerioPatch (Izun Pharmaceuticals Corp, New York, NY) is a herbal patch designed to reduce the signs and symptoms of inflammation caused by gingivitis and periodontitis. The patch has two layers composed of a nonabsorptive matrix, which allows for slow dissolution of the inner layer. This adjunct provides a protective seal over inflamed gingival and oral mucosa, which creates a barrier, provides relief, and promotes wound healing by absorbing the local inflammatory exudate from the inflamed tissue.30
Recognition that elevations in pro-inflammatory mediators were driving the periodontal disease process resulted in the development of an FDA-approved host modulatory therapy known as Periostat. This approach evolved as a new use for an existing drug, doxycycline, which, when used at a subantimicrobial dose (20 mg) could inhibit the destructive enzymes and reduce excessive levels of cytokines. Tested in multiple randomized clinical trials, this approach was found to be an effective adjunct to scaling and root planing, leading to significant improvements in probing depth reductions, clinical attachment level gains, and reductions in bleeding on probing.31 It also has been shown to boost the effects of LAAs.32 Clinical studies have supported its use in high-risk patients who can be difficult to manage, such as smokers,33 individuals with diabetes34 and women with osteoporosis/osteopenia,35 as well as those at risk for cardiovascular disease.36 Available in generic form by prescription, it has been shown to be safe and effective in up to two years of continuous use in a study of osteopenic women.37 It should not be used in patients with allergies to tetracyclines, pregnant or lactating women, or children younger than 12.
CONCLUSION
In light of the foregoing discussion, treatments that combine mechanical therapies and adjunctive chemotherapeutics enable clinicians to better manage gingivitis and periodontitis. The use of antiseptics, antibiotics and host modulatory therapy as adjuncts to brushing, ultrasonics, scaling and root planing have made nonsurgical therapies more predictable. Faced with multiple options, the provider must ultimately consider the evidence when developing treatment strategies that address the unique needs of each patient.
Rebecca S. Wilder, RDH, MS, has no commercial conflicts of interest to disclose. Maria Emanuel Ryan, DDS, PhD, is vice president and chief dental officer for Colgate-Palmolive Co.
References
- Kepic TJ, O’Leary TJ, Kafrawy AH. Total calculus removal: an attainable objective? J Periodontol. 1990;61:16.
- Listgarten MA. Structure of the microbial flora associated with periodontal health and diseases in man. J Periodontol. 1976;47:1–18.
- Cobb CM, Killoy WJ. Microbial colonization in human periodontal disease: an illustrated tutorial on selected ultrastructural and ecologic considerations. Scan Microsc. 1990;4:675–691.
- Shiau HJ, DePaola LG. Chemotherapeutic agents. In: Spolarich AE, Panagakos FS. Prevention Across the Lifespan: A Review of Evidence-Based Interventions for Common Oral Conditions. Charlotte, NC: Professional Audience Communications; 2017.
- Elworthy A, Greenman J, Doherty FM, et al. The substantivity of a number of oral hygiene products determined by the duration of effects on salivary bacteria. J Periodontol. 1996;76:572–576.
- Canton J, Ryan ME. Clinical studies on the management of periodontal diseases utilizing subantimicrobial dose doxycycline (SDD). Pharmacol Res. 2011;63:114–120.
- Gunsolley JC. A meta-analysis of six-month studies of antiplaque and antigingivitis agents. J Am Dent Assoc. 2006;137:1649–1657.
- Van Strydonck DA, Slot DE, Van der Velden U, et al. Effect of a chlorhexidine mouthrinse on plaque, gingival inflammation and staining in gingivitis patients: a systematic review. J Clin Periodontol. 2012;39:1042–1055.
- Addy M. Oral hygiene products: potential for harm to oral and systemic health? Periodontol 2000. 2008;48:54–65.
- Wilder RS, Bray KS. Improving periodontal outcomes: merging clinical and behavioral science. Periodontol 2000. 2016;71:65–81.
- Addy M. Chlorhexidine compared with other locally delivered antimicrobials. J Clin Periodontol. 1986;13:957–964.
- Stoeken JE, Paraskevas S, van der Weijden GA. The long-term effect of a mouthrinse containing essential oils on dental plaque and gingivitis: a systematic review. J Periodontol. 2007;78:1218–1228.
- Haps S, Slot DE, Berchier CE, Van der Weijden GA. The effect of cetylpyridinium chloride-containing mouth rinses as adjuncts to toothbrushing on plaque and parameters of gingival inflammation: a systematic review. Int J Dent Hyg. 2008;6:290–303.
- Volpe AR, Petrone ME, DeVizio W, et al. A review of plaque, gingivitis, calculus and caries clinical efficacy with a dentifrice containing triclosan and PVM/MA Copolymer. J Clin Dent. 1993;4:31–41.
- Davies RM, Ellwood RP, Davies GM. The effectiveness of a toothpaste containing triclosan and polyvinyl-methyl ether maleic acid copolymer in improving plaque control and gingival health: a systematic review. J Clin Periodontol. 2004:31:1029–1033.
- Hioe KP, van der Weijden GA. The effectiveness of self-performed mechanical plaque control with triclosan containing dentifrices. Int J Dent Hyg. 2005;3:192–204.
- Haraszthy VI, Sreenivasan PK, Zambon JJ. Community-level assessment of dental plaque bacteria susceptibility to triclosan over 19 years. BNC Oral Health. 2014;2:14–61.
- Riley P, Lamont T. Triclosan/copolymer containing toothpastes for oral health. Cochrane Database Syst Rev. 2013;5:12.
- Federal Register, Vol. 78, No. 242, December 17, 2013. Available at: https://www.gpo.gov/fdsys/pkg/FR-2013-12-17/pdf/2013-29936.pdf. Accessed March 13, 2018.
- Cullinan MP, Bird PS, Heng NC, West MJ, Seymour GJ. No evidence of triclosan-resistant bacteria following long-term use of triclosan-containing toothpaste. J Periodontal Res. 2014;49:220–225.
- Jeffcoat MK, Bray KS, Ciancio SG, et al. Adjunctive use of a a subgingival controlled-release chlorhexidine chip reduces probing depth and improves attachment level compared with scaling and root planing alone. J Periodontol. 1998;69:989–997.
- Stoller NH, Johnson LR, Trapnell S, et al. The pharmacokinetic profile of a biodegradable controlled release delivery system containing doxycycline compared to systemically delivered doxycycline in gingival crevicular fluid, saliva, and serum. J Periodontol. 1998;69:1085–1091.
- Christersson LA. Tissue response and release of minocycline after subgingival deposition by use of a resorbable polymer. Warminster, Pa: OraPharma Inc; 1988.
- Goodson JM, Gunsolley JC, Grossi SG, et al. Minocycline HCI microspheres reduce red-complex bacteria in periodontal disease therapy. J Periodontol. 2007;78:1568–1579.
- Machion L, Andia DC, Lecio G, et al. Locally delivered doxycycline as an adjunctive therapy to scaling and root planing in the treatment of smokers: a 2-year follow-up. J Periodontol. 2006;77:606–613.
- D’Aiuto F, Parkar M, Nibali L, et al. Periodontal infections cause changes in traditional and novel cardiovascular risk factors: results from a randomized controlled clinical trial. Am Heart J. 2006;151:977–984.
- Tonetti MS, D’Aiuto F, Nibali L, et al. Treatment of periodontitis and endothelial function. N Engl J Med. 2007;356:911–920.
- Smiley CJ, Tracy SL, Abt E, et al. Systematic review and meta-analysis on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:508–524.
- Smiley CJ, Tracy SL, Michalowicz BS, et al. Evidence–based clinical practice guideline on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc. 2015;146:525–535.
- Chaushu L, Weinreb M, Beitlitum I, et al. Evaluation of a topical herbal patch for soft tissue wound healing: an animal study. J Clin Periodontol. 2015;42:288–293.
- Ciancio S, Ashley R. Safety and efficacy of sub-antimicrobial-dose doxycycline therapy in patients with adult periodontitis. Adv Dent Res. 1998;12:27–31.
- Novak MJ, Dawson DR 3rd, Magnusson I, et al. Combining host modulation and topical antimicrobial therapy in the management of moderate to severe periodontitis: a randomized multicenter trial. J Periodontol. 2008;79:33–41.
- Preshaw PM, Hefti AF, Bradshaw MH, et al. Adjunctive subantimicrobial dose doxycycline in smokers and non-smokers with chronic periodontitis. J Clin Periodontol. 2005;32:610–616.
- Martorelli de Lima AF, Cury CC, Bazan Paliota D, et al. Therapy with adjunctive doxycycline local delivery in patients with type 1 diabetes mellitus and periodontitis. J Clin Periodontol. 2004;31:648–653.
- Reinhardt RA, Stoner JA, Golub LM, et al. Efficacy of sub-antimicrobial dose doxycycline in post-menopausal women: clinical outcomes. J Clin Periodontol. 2007;34:768–775.
- Brown DL, Desai KK, Vakili BA, et al. Clinical and biochemical results of the metalloproteinase inhibition with subantimicrobial doses of doxycycline to prevent acute coronary syndromes (MIDAS) pilot trial. Arterioscler Thromb Vasc Biol. 2004;24:733–738.
- Walker C, Puumala S, Golub LM, et al. Subantimicrobial dose doxycycline effects on osteopenic bone loss: microbiologic results. J Periodontol. 2007;78:1590–1601.
Featured image by CLICK_AND_PHOTO/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. April 2018;4(4):49-52.




