
CE Sponsored by Dentsply Sirona Midwest: Dental Handpiece Care and Processing
Following recommended methods for cleaning, maintaining and sterilizing handpieces can provide a safe care environment for patients and staff.
PURCHASE COURSE
This course was published in the February 2017 issue and expires February 2020. The author has no commercial conflicts of interest to disclose. This 2 credit hour self-study activity is electronically mediated.
OBJECTIVES
After reading this course, the participant should be able to:
- Discuss disease transmission in dental settings.
- Describe when federal infection prevention guidelines were introduced, the nature of the recommendations, and the basic principles of infection control.
- List U.S. Centers for Disease Control and Prevention (CDC) dental handpiece sterilization guidelines.
- Explain the need to consider recommendations from CDC, as well as device manufacturers, when cleaning, maintaining and sterilizing handpieces.
INTRODUCTION
Is your office in compliance with current U.S. Centers for Disease Control and Prevention (CDC) guidelines for processing high-speed and low-speed handpieces? According to CDC estimates, 2.7 to 3.9 million Americans are living with chronic hepatitis C, and, in 2009, an estimated 16,000 new hepatitis C viral infections occurred.* These figures underscore the importance of following accepted infection prevention protocols in dental practice.
Made possible through an unrestricted educational grant from Dentsply Sirona Midwest, this continuing education article is designed to help practices comply with CDC guidelines, and establish a documented training record for office staff. Ensuring that each team member processes instruments according to approved guidelines is a critical step in protecting staff and patients from disease transmission. Dentsply Sirona is thankful to Shellie Kolavic Gray, DMD, MPH, for authoring this insightful paper supporting the practice of safe dentistry.
Jonathan Whiteside
Midwest Brand Product Manager
Dentsply Sirona
Preventing or controlling the transmission of disease is the foundation of safe dental practice. Compared to hospital-acquired infections, disease transmission in dental settings is relatively rare.1,2 When transmissions occur, however, practices are scrutinized to determine whether infection control efforts were incomplete, incorrect or absent. In most — if not all — cases, investigators have determined that transmission was preventable if clinicians had followed infection control guidelines recommended by the U.S. Centers for Disease Control and Prevention (CDC).3–5 These guidelines and recommendations are developed to optimize health care and prevent disease, injury or other threats to public health. Guidelines for implementing recommended infection control practices in dental settings were first published by CDC in 1986, and updated or summarized in 1993, 2003 and 2016.6–9
Dental handpieces are contaminated during patient treatment through contact with mucous membranes, oral fluids and other potentially infective patient materials. For several decades, handpieces were considered too fragile to withstand the rigors of sterilization, and external cleaning and disinfection was standard practice.6 In 1986, CDC’s first guideline document for dental infection control acknowledged that many handpieces in use at that time could be sterilized, but stated only that routine sterilization was desirable.8 In addition to the obvious contamination of external surfaces during use, research demonstrated that patient material can be drawn into the internal components of handpieces.8,10–14 The results of these studies indicated that unless all components were completely sterilized between patients, debris and microorganisms that were drawn into dental handpieces could be released during treatment of subsequent patients.
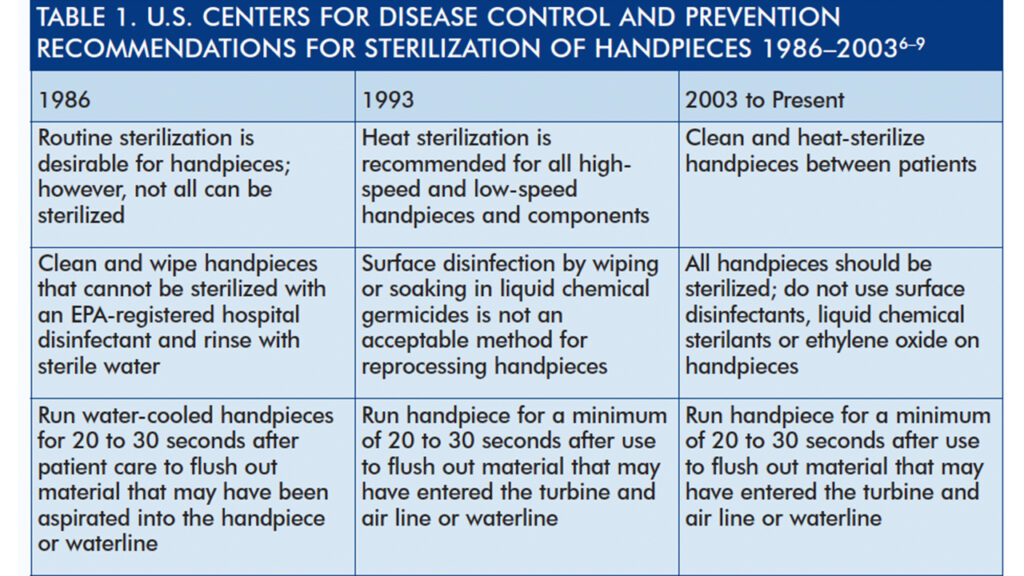 With the introduction of updated, evidence-based guidelines in 2003, CDC firmly recommended heat sterilization for all handpieces, component parts (including low-speed motors), and attachments, such as reusable prophy angles8 (Table 1).
With the introduction of updated, evidence-based guidelines in 2003, CDC firmly recommended heat sterilization for all handpieces, component parts (including low-speed motors), and attachments, such as reusable prophy angles8 (Table 1).
CDC’s Guidelines for Infection Control in Dental Health-Care Settings — 2003 represents the current standard of care for dental infection control.8 In 2016, CDC relaunched the infection control guidelines to a new generation of dental professionals with its Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Dental Care9 (Table 2). Although it presents new information and a few updated guidelines, the 2016 summary is intended to be a companion to the 2003 document, not a replacement. Because the principles of infection control have not changed, the 2003 document continues to be the standard for infection control in dental settings. Individuals who seek more information about the science supporting each recommendation should consult the original 2003 document.8 The 2003 and 2016 documents are available at cdc.gov/oralhealth/infectioncontrol/guidelines/.
![table-2]() PRINCIPLES OF INFECTION CONTROL
PRINCIPLES OF INFECTION CONTROL
Dental infection control practices align with four principles that represent the most basic actions for preventing disease transmission. These principles drive the critical procedures for making dental visits safe for both patients and providers.15
- Take Action to Stay Healthy: Obtaining recommended immunizations against hepatitis B, tetanus and the flu virus are examples of taking action to stay healthy, especially if an exposure injury occurs while handling dental handpieces.16
- Avoid Contact with Blood and Body Fluids: Handpieces regularly contact saliva and blood during dental treatment. CDC’s infection control recommendations focus on preventing direct contact with these and other potentially infectious materials.
- Limit the Spread of Blood and Body Fluid Contamination: In addition to preventing direct contact, limiting the spread of blood and body fluids is key to preventing disease transmission.
- Make Objects Safe for Use: Proper cleaning, disinfection and sterilization ensure that patient care items are not only safe for dental personnel to handle, but also for use on subsequent patients.
Although dental handpieces have not been solely implicated in transmission of disease, the internal surfaces of all handpieces — whether high-speed, low-speed or electric — are contaminated to some degree after use. While CDC does not detail individual steps for handpiece sterilization, its recommended actions for infection control apply to all areas of dental practice, including processes for making handpieces safe for reuse.8,9 The basic steps for cleaning and preparing handpieces for sterilization are similar for virtually all types of handpieces. That noted, each manufacturer provides specific directions for their particular devices. These instructions should not be considered optional, and noncompliance with the recommended practices outlined by the manufacturer may damage the device and/or void the warranty for the handpiece or component parts.
STAFF TRAINING AND PREPARATION
Processing handpieces for reuse requires detailed knowledge of the manufacturer’s instructions for cleaning, maintenance and sterilization. Dental teams must also know how to safely use cleaning products, and follow effective infection control methods. Handpieces should never be cleaned, maintained or sterilized without having the manufacturer’s instructions, nor should these tasks be performed by untrained staff. CDC recommends training all dental personnel in the basic principles of infection control and preventing pathogenic transmission.8,9 In addition, CDC recommends providing job-specific or task-specific infection control training to all dental personnel who perform duties beyond that required by the federal Occupational Safety and Health Administration’s (OSHA) bloodborne pathogen standard.8,9,17
Handling contaminated instruments, including a handpiece of any kind, requires clear understanding of the principles of infection control and the manufacturer’s instructions for use. Dental handpieces are medical devices regulated by the U.S. Food and Drug Administration (FDA). Manufacturers of reusable medical or dental devices are responsible for labeling that provides adequate directions for use and instructions for preparing the device for patient reuse.
The FDA lists six criteria for reprocessing instructions,18 and specifies each device should have directions that:
- Reflect the physical design of the device, its intended use, and the contamination expected during clinical use
- Clearly advise users to thoroughly clean the instrument
- Indicate the appropriate cleaning, disinfection or sterilization processes for the device
- Are technically feasible and performed with legally marketed devices and accessories
- Are understandable and written in plain language
- Are comprehensive, with steps that describe:
- Prompt, initial surface-cleaning steps to prevent debris from drying prior to final cleaning
- Disassembly and reassembly of devices with moving parts to facilitate cleaning
- Detailed cleaning processes using validated manual or mechanical methods
- Effective cleaning agents compatible with the device
- Rinsing after cleaning
- Lubricating agents that are compatible with the device
- Visual inspection methods to determine whether the device appears clean and undamaged
- Sterilization methods that follow the sterilizer manufacturer’s instructions for use
CDC recommends that manufacturer instructions for reprocessing reusable dental instruments and equipment be readily available — ideally in or near the reprocessing area. CDC also recommends that dental practices inform all staff of OSHA guidelines for exposure to chemical agents used for disinfection and sterilization.8,9
PERSONAL PROTECTIVE EQUIPMENT
The handpiece cleaning process requires handling of a patient care item that is contaminated inside and out. When contact is likely with contaminants that include blood, saliva or other potentially infectious material, acceptable personal protective equipment must be available. CDC’s key recommendations for personal protection include the appropriate use of protective clothing, gloves, masks and eye protection; in other words, items that provide a barrier from direct contact with contaminants.8,9
FLUSHING AND CLEANING
In addition, CDC recommends that any device used in the patient’s mouth that is connected to the office’s air/water system should be run for 20 to 30 seconds to flush the handpiece and help expel patient material that may remain in the handpiece, and air line or waterline.8,9 After flushing, the handpiece should be cleaned in preparation for sterilization. Some manufacturers may recommend cleaning the handpiece at the point of patient use. CDC recommends that each practice designate a central processing area with distinct areas for:8,9
- Receiving, cleaning and decontamination
- Preparation and packaging
- Sterilization
- Storage of patient care items
CDC also recommends following safe practices to minimize exposure or injury by transporting these items to the processing area in a covered container.8,9 Holding solutions, while recommended for temporary storage of contaminated instruments, may not be appropriate for handpieces; in fact, manufacturers may warn against immersion of handpieces and components. Furthermore, CDC does not recommend use of liquid chemical sterilants or high-level disinfectants as holding solutions for instruments or equipment. In addition to the personal protective equipment previously discussed, the use of puncture- and chemical-resistant utility gloves is warranted when processing handpieces or other instruments.8,9
There are no distinct CDC guidelines that suggest how to clean specific handpieces; as noted, cleaning protocols should follow the manufacturer’s instructions and CDC infection control recommendations. Dental teams should attempt to clean all visible blood and other contamination from instruments and devices prior to sterilization or disinfection procedures. Whether visible or not, dried saliva, blood or other potentially infectious material are present on a used handpiece. If not removed, debris can serve as insulation that protects pathogens from the lethal environment inside the sterilizer. Manufacturers may recommend cleaning the handpiece under running water, with alcohol, or with disinfectant wipes, and it is important to follow the instructions in order to avoid damage.
Although CDC recommends automated cleaning equipment to remove debris from dental instruments and decrease staff exposure to blood, handpiece manufacturers generally do not advise ultrasonic cleaning or immersion in chemical disinfectants.8 Although some handpieces and components can be placed in medical-grade instrument washers or washer-disinfectors, dishwashers are not FDA approved for cleaning dental instruments and may damage handpieces.
HANDPIECE LUBRICATION
Lubricating dental handpieces can be a challenge for a busy office, but, when indicated, this is a vital step in handpiece maintenance. Specialized lubricants are used for handpieces and components, and should be properly applied in the correct location and quantity directed by the manufacturer. Although CDC does not offer specific recommendations for handpiece lubrication, it is important to note that used handpieces are contaminated, and when lubricant spray nozzles are inserted into handpieces, the nozzles also become contaminated.
Clinicians should consult the manufacturer regarding ways to prevent or minimize cross-contamination during lubrication procedures, including expelling excess lubricant from the device. After applying lubricant, the instructions may state that handpieces and attachments should be reconnected to the unit or another air supply to distribute the lubricant and expel excess material. If a dedicated air supply is not located in the instrument preparation room, it will be necessary for staff to return to the operatory and reattach the handpiece to the dental unit. This raises several infection control considerations; for example, the operatory may be already cleaned or in use, and expelling lubricant in the operatory or an adjacent room may cause cross-contamination. Some handpieces require lubrication after sterilization. This poses an infection control challenge because it breaks the sterility cycle. If a handpiece requires lubrication after sterilization, consult the manufacturer to determine the best course of action.
AUTOMATED MAINTENANCE
Automated handpiece maintenance systems are available, and while individual units vary, they are designed to save staff time by performing internal cleaning and lubrication. Before purchasing an automated handpiece maintenance system, carefully review its features and functionality. Automated maintenance systems may provide internal cleaning only, in which case handpiece exteriors must be thoroughly cleaned prior to being placed in the maintenance unit. In addition, the handpieces are not yet sterile and should be handled carefully while wearing gloves and other appropriate personal protective equipment.
STERILIZATION PROCESSING
Once a handpiece is cleaned and lubricated, it is still not ready for patient reuse until it undergoes sterilization. It is the dental team’s responsibility to ensure the process is compatible with the handpiece and components to be sterilized. CDC states that surface disinfection, immersion in chemical germicides, or use of ethylene oxide gas are not acceptable or effective methods for sterilizing handpieces or components.8
All sterilization procedures should be completed with medical sterilization equipment cleared by the FDA. CDC recommends heat sterilization for handpieces,8,9 but, again, consult the manufacturer for specifics. Handpieces and attachments should be prepared, and wrapped or packaged for sterilization, as directed by the handpiece manufacturer and the packaging material and sterilizer unit manufacturers. In addition, CDC recommends labeling processed items, noting the sterilizer used, date of sterilization, the cycle or load number, and, if applicable, expiration date.8,9
STERILIZING UNWRAPPED HANDPIECES
The routine sterilization of unwrapped patient care items is not recommended by CDC.8 Formerly called “flash sterilization,” this method is now termed “immediate-use sterilization” to highlight that the item is to be used promptly when removed from the sterilizer after cooling and not stored for future use. If the device was sterilized while unwrapped, however, the newly sterile item must be protected from contamination during transport to the point of patient use. All practices should have a carefully prepared, written protocol for minimizing the risk of contaminating unwrapped instruments that have undergone immediate-use sterilization. CDC discourages storage, even temporary, of unwrapped instruments that have undergone immediate use sterilization because they can quickly become contaminated.8,9
When immediate-use sterilization is needed, CDC outlines certain conditions that must be met, including:8
- The need to thoroughly clean and dry instruments prior to the unwrapped sterilization cycle
- Use of mechanical and chemical indicators for each unwrapped sterilization cycle
- Allowing unwrapped instruments to dry and cool in the sterilizer before they are handled to avoid contamination and thermal injury
- Handling instrument aseptically during removal from the sterilizer, and while transporting to the point of use
MONITORING PROTOCOLS
Of course, proper cleaning, lubrication and packaging is ineffective if the sterilizer is not able to reach the conditions needed to render items safe for patient use. Therefore, CDC guidelines recommend using a combination of mechanical, chemical and biological monitoring to evaluate the conditions of the sterilizer and effectiveness of the sterilization procedure. Mechanical and chemical indicators should be used with every sterilization cycle to help detect equipment malfunction and procedural errors.8,9 Mechanical monitoring includes assessing the parameters of cycle time, temperature and pressure by visually observing the gauges or displays on the sterilizer unit. CDC recommends monitoring and recording the mechanical aspects of the sterilizer for each processed load.8,9 Although correct readings do not ensure that complete sterilization of handpieces and other instruments has occurred, incorrect readings can be an early signal of a problem with the sterilization cycle.
Internal and external chemical indicators contain sensitive inks that change color according to the physical conditions inside the sterilizer during processing. External chemical indicators are affixed to the outside of a package, and a color change verifies the package has been exposed to the sterilization process. Internal chemical indicators signal the sterilizing agent has penetrated the package. It is important note that neither external nor internal indicators prove that sterilization has been achieved — however, they help detect malfunctions or procedural errors during the sterilization cycle. When this occurs, it is advisable to thoroughly check the equipment and repeat the sterilization cycle.8
The weekly use of biological indicators (e.g., spore tests) is recommended to monitor the sterilization process.8,9 Biological indicators are the most widely accepted method because they directly assess sterilization by sending highly resistant bacteria spores through a complete sterilization cycle. Compared to common microbial contaminants found on handpieces and other dental equipment, the spores used for biological indicators are more resistant to sterilization procedures. If test spores are killed during the cycle, other potential pathogens in the load have been killed, as well. If the sterilization cycle does not kill the spores, however, the sterilizer should immediately be removed from service, and further investigation is warranted. CDC’s recommended protocol following a spore-test failure is available at cdc.gov/ oralhealth/ infection control/ faq/ sterilization_ monitoring.htm. All practices should maintain sterilizing equipment regularly, and keep accurate sterilizer maintenance records. Sterilization processing should only be assigned to staff who are thoroughly trained to perform the essential steps and respond appropriately to possible sterilization failures.
SUMMARY
Complying with guidelines for handpiece cleaning, maintenance and sterilization requires dental teams to be knowledgeable of recommended infection control practices and manufacturer’s instructions for use. Initial and ongoing training, as recommended by CDC, is available through textbooks and self-training materials, continuing education articles and seminars, infection control consultants, and dental infection control professional organizations, such as the Organization for Safety, Asepsis and Prevention.19–21
The purchase, maintenance, processing and replacement of handpieces and attachments represent a significant investment for a dental practice. Following safe methods for cleaning, maintenance and sterilization should maximize the life of handpieces, while providing a safe care environment for patients and staff.
*National Center for HIV/AIDS, Viral Hepatitis, STD and TB Prevention. Disease Burden from Viral Hepatitis A, B, and C in the United States. Available at: cdc.gov/hepatitis/pdfs/disease_ burden.pdf. Accessed January 18, 2017.
REFERENCES
- U.S. Centers for Disease Control and Prevention. 2014 National and State Healthcare-Associated Infections Progress Report. Available at: cdc.gov/hai/progress-report/index.html. Accessed February 16, 2017.
- Cleveland JL, Gray SK, Harte JA, Robison VA, Moorman AC, Gooch BF. Transmission of blood-borne pathogens in U.S. dental health care settings: 2016 update. J Am Dent Assoc. 2016;147:729–738.
- Redd JT, Baumbach J, Kohn W, Nainan O, Khristova M, Williams I. Patient-to-patient transmission of hepatitis B virus associated with oral surgery. J Infect Dis. 2007;195:1311–1314.
- Radcliffe RA, Bixler D, Moorman A, et al. Hepatitis B virus transmissions associated with a portable dental clinic, West Virginia, 2009. J Am Dent Assoc. 2013;144:1110–1118.
- Peralt G, Tobin-D’Angelo M, Parham A, et al. Notes from the field: Mycobacterium abscessus infections among patients of a pediatric dentistry practice — Georgia, 2015. MMWR Morb Mortal Wkly Rep. 2016;65:355–356.
- U.S. Centers for Disease Control and Prevention. Recommended infection-control practices for dentistry. MMWR Morb Mortal Wkly Rep. 1986;35:237–242.
- U.S. Centers for Disease Control and Prevention. Recommended infection-control practices for dentistry, 1993. MMWR Morb Mortal Wkly Rep. 1993;42:RR-8.
- U.S. Centers for Disease Control and Prevention. Guidelines for infection control in dental health-care settings — 2003. MMWR Morb Mortal Wkly Rep. 2003;52:1–66.
- U.S. Centers for Disease Control and Prevention. Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care. Available at: cdc.gov/oralhealth/infectioncontrol/pdf/safe-care2.pdf. Accessed February 16, 2017.
- Crawford JJ, Broderius C. Control of cross-infection risks in the dental operatory: prevention of water retraction by bur cooling spray systems. J Am Dent Assoc. 1988;116:685–687.
- Lewis DL, Boe RK. Cross-infection risks associated with current procedures for using high-speed dental handpieces. J Clin Microbiol. 1992;30:401–406.
- Mills SE, Kuehne JC, Bradley DV Jr. Bacteriological analysis of highspeed handpiece turbines. J Am Dent Assoc. 1993;124:59–62.
- Epstein JB, Rea G, Sibau L, Sherlock CH, Le ND. Assessing viral retention and elimination in rotary dental instruments. J Am Dent Assoc. 1995;126:87–92.
- Checchi L, Montebugnoli L, Samaritani S. Contamination of the turbine air chamber: a risk of cross infection. J Clin Periodontol. 1998;25:607–611.
- U.S. Centers for Disease Control and Prevention. Practical Infection Control in the Dental Office. U.S. Department of Health and Human Services, Public Health Service. 1993.
- U.S. Centers for Disease Control and Prevention. Immunization of Health-Care Personnel: Recommendations of the Advisory Committee on Immunization Available at: cdc.gov/mmwr/pdf/rr/rr6007.pdf. Accessed February 16, 2017.
- Occupational Safety & Health Administration. Bloodborne Pathogens and Needlestick Prevention Standards. Available at: osha.gov/SLTC/ bloodbornepathogens/index.html. Accessed February 16, 2017.
- U.S. Food and Drug Administration. Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling. Available at: fda.gov/downloads/medicaldevices/deviceregulationandguidance/guidance documents/ucm253010.pdf. Accessed February 16, 2017.
- Miller CH, Palenik DJ, eds. Infection Control and Management of Hazardous Materials for the Dental Team. 6th ed. St. Louis: Elsevier; 2017.
- Organization for Safety, Aspesis and Prevention (OSAP). From Policy to Practice: OSAP’s Guide to the CDC Guidelines. Available at: osap.org/?CDCGuidelinesCourse. Accessed February 16, 2017.
- Organization for Safety, Aspesis and Prevention. OSHA and CDC Guidelines. Interact Training System. Available at: osap.org/?page=InteractProgram. Accessed February 16, 2017.
Featured photo by ABEZIKUS/ISTOCK/GETTY IMAGES PLUS
From Decisions in Dentistry. February 2017;3(2):45–50.





